order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Locations
Orders Processing,
Shipping & Receiving,
Warehouse
2 Shaker Rd Suites
B001/B101
Shirley, MA 01464
Production Lab
Floor 6, Suite 620
20700 44th Avenue W
Lynnwood, WA 98036
Telephone Numbers
Tel: +1 (206) 374-1102
Fax: +1 (206) 577-4565
Contact Us
Additional Contact Details
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
order histories, retained contact details for faster checkout, review submissions, and special promotions.
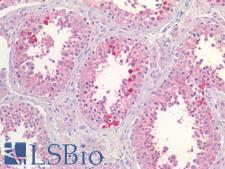
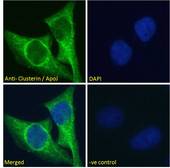
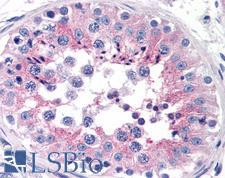
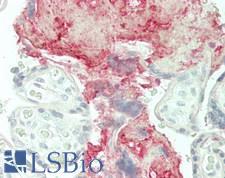
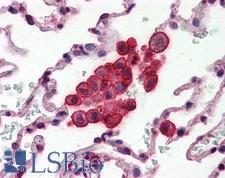
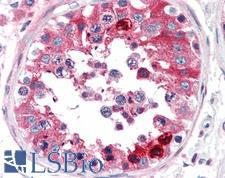
CLU / Clusterin
clusterin
Isoform 1 functions as extracellular chaperone that prevents aggregation of nonnative proteins. Prevents stress-induced aggregation of blood plasma proteins. Inhibits formation of amyloid fibrils by APP, APOC2, B2M, CALCA, CSN3, SNCA and aggregation-prone LYZ variants (in vitro). Does not require ATP. Maintains partially unfolded proteins in a state appropriate for subsequent refolding by other chaperones, such as HSPA8/HSC70. Does not refold proteins by itself. Binding to cell surface receptors triggers internalization of the chaperone-client complex and subsequent lysosomal or proteasomal degradation. Secreted isoform 1 protects cells against apoptosis and against cytolysis by complement. Intracellular isoforms interact with ubiquitin and SCF (SKP1-CUL1-F-box protein) E3 ubiquitin-protein ligase complexes and promote the ubiquitination and subsequent proteasomal degradation of target proteins. Promotes proteasomal degradation of COMMD1 and IKBKB. Modulates NF-kappa-B transcriptional activity. Nuclear isoforms promote apoptosis. Mitochondrial isoforms suppress BAX-dependent release of cytochrome c into the cytoplasm and inhibit apoptosis. Plays a role in the regulation of cell proliferation.
| Gene Name: | clusterin |
| Synonyms: | CLU, APO-J, Apolipoprotein J, CLU1, Clusterin, Complement lysis inhibitor, CLI, AAG4, Aging-associated protein 4, Ku70-binding protein 1, KUB1, SGP2, APOJ, SGP-2, Sulfated glycoprotein 2, CLU2, Complement cytolysis inhibitor, NA1/NA2, SP-40 |
| Target Sequences: | NM_001831 NP_001822.3 P10909 |
Publications (4)



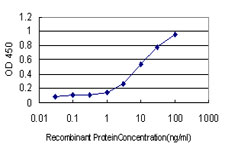
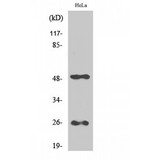
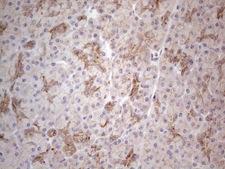
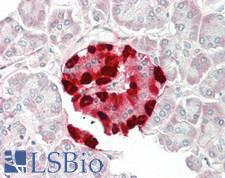
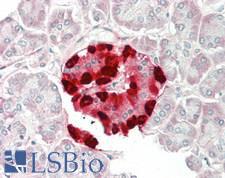
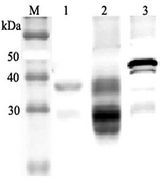
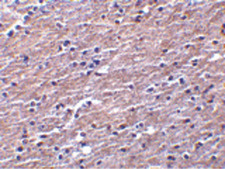
If you do not find the reagent or information you require, please contact Customer.Support@LSBio.com to inquire about additional products in development.










