order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Locations
Orders Processing,
Shipping & Receiving,
Warehouse
2 Shaker Rd Suites
B001/B101
Shirley, MA 01464
Production Lab
Floor 6, Suite 620
20700 44th Avenue W
Lynnwood, WA 98036
Telephone Numbers
Tel: +1 (206) 374-1102
Fax: +1 (206) 577-4565
Contact Us
Additional Contact Details
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Retroviridae (HIV)
Retroviruses belong to a family of enveloped RNA viruses (Retroviridae) that infect vertebrates. Retroviruses are divided into five sub genera, with two members (Delta retroviruses HTLV-1 and HTLV-2 and Lentiviruses, HIV-1 and HIV-2) causing human disease. They can be exogenous, transmitted horizontally among hosts, or endogenous, inherited vertically through the genomes of their hosts. The virus requires a reverse transcriptase to convert the viral RNA genome into DNA, which integrates into host chromosomes and utilizes host proteins for gene expression and replication. Retroviruses can also be oncogenic through insertional activation of host genes. In humans, HIV is responsible for causing the acquired immunodeficiency syndrome (AIDS) and HTLV-1 and HTLV-2 are the retroviruses that cause human T-cell leukemia/lymphomas.
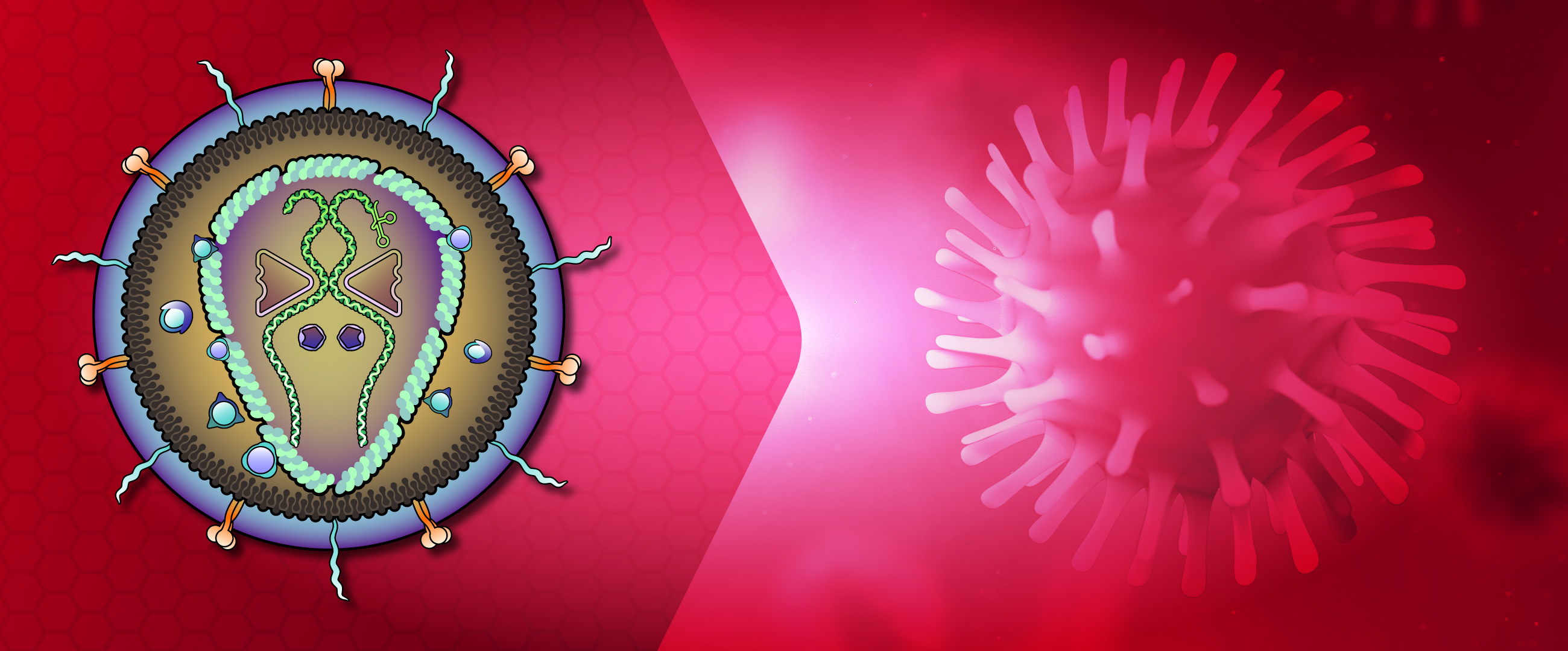
About Retroviridae
Structurally, retroviruses are enveloped viruses with an RNA genome. The virion contains a highly error-prone RNA-dependent DNA Polymerase (reverse transcriptase) that converts the viral RNA into DNA, which integrates into the host chromosome (Hu, 2012). They have a diploid genome, with 2 copies of RNA per viral particle. A lentiviral (HIV) genome is depicted here.
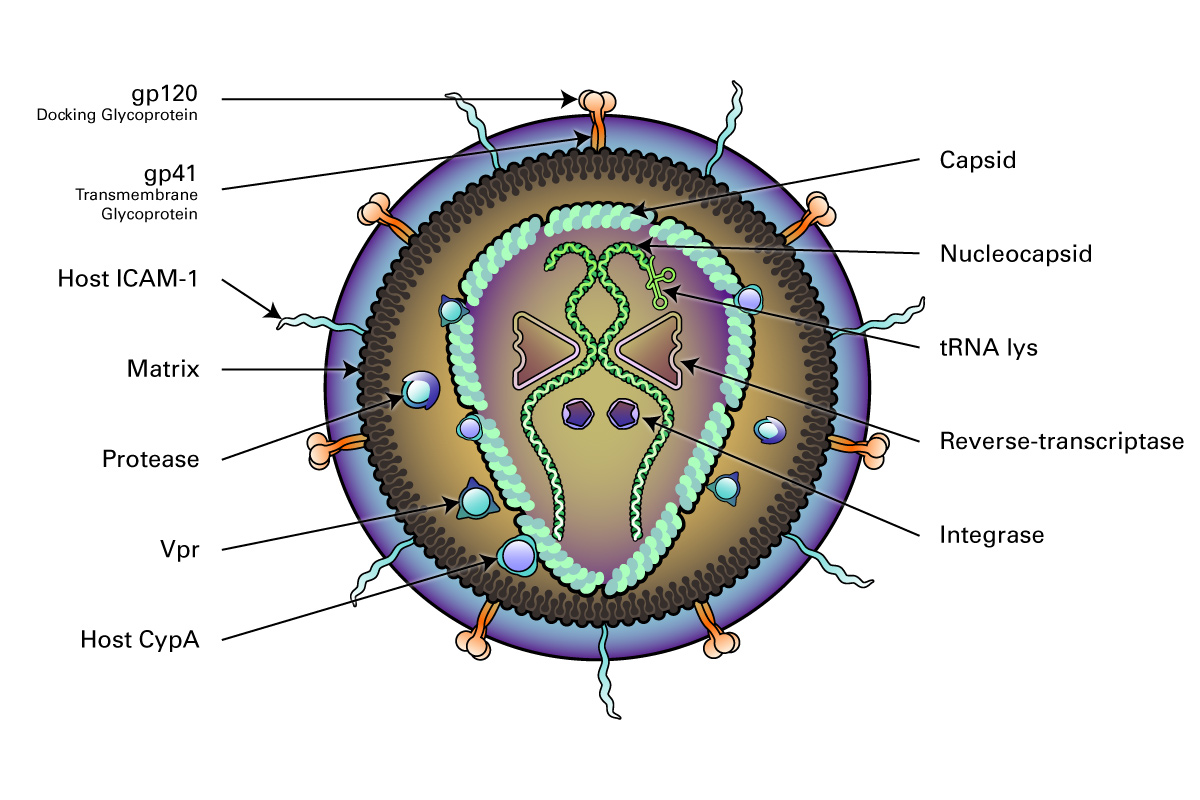
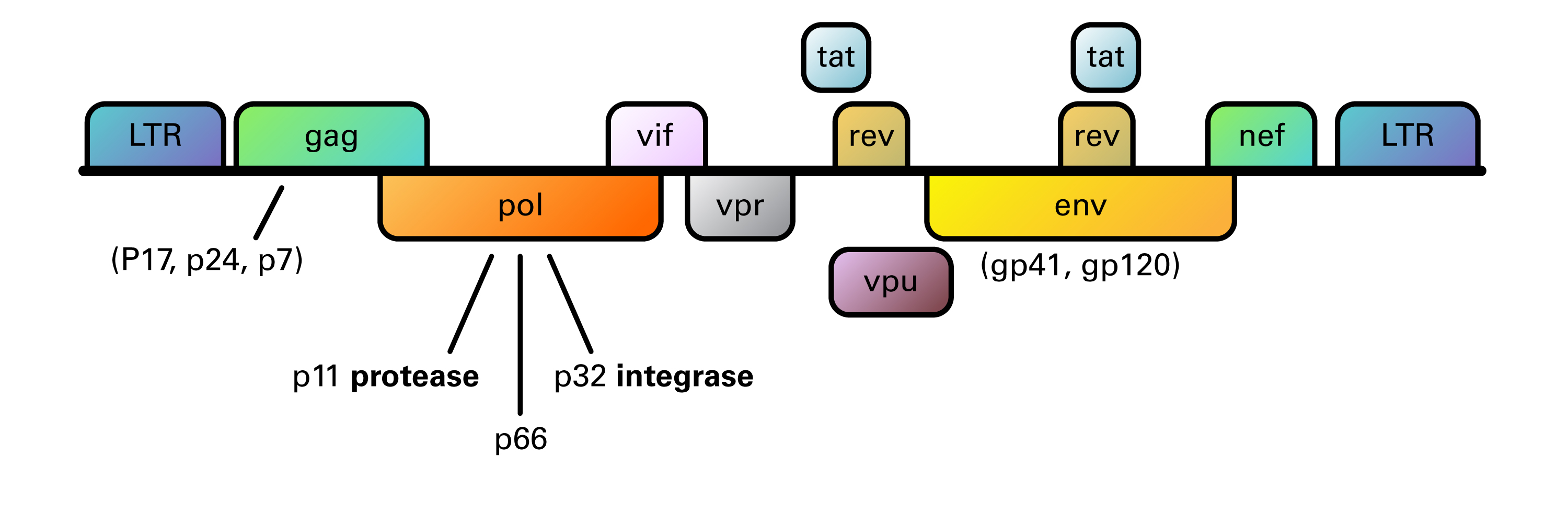
Organization of the HIV-1 genome showing the reading frames of the genes coding for structural and regulatory proteins: LTR = long terminal repeat; gag = group-specific antigen; pol = polymerase; env = envelope. The gag, pol, and env genes are expressed as precursor polyproteins, which are then cleaved to yield mature viral proteins. In the case of the regulator genes, the proteins of tat and rev are composed of two gene regions. The genome consists of 9,200-9,600 nucleotides in HIV-1.
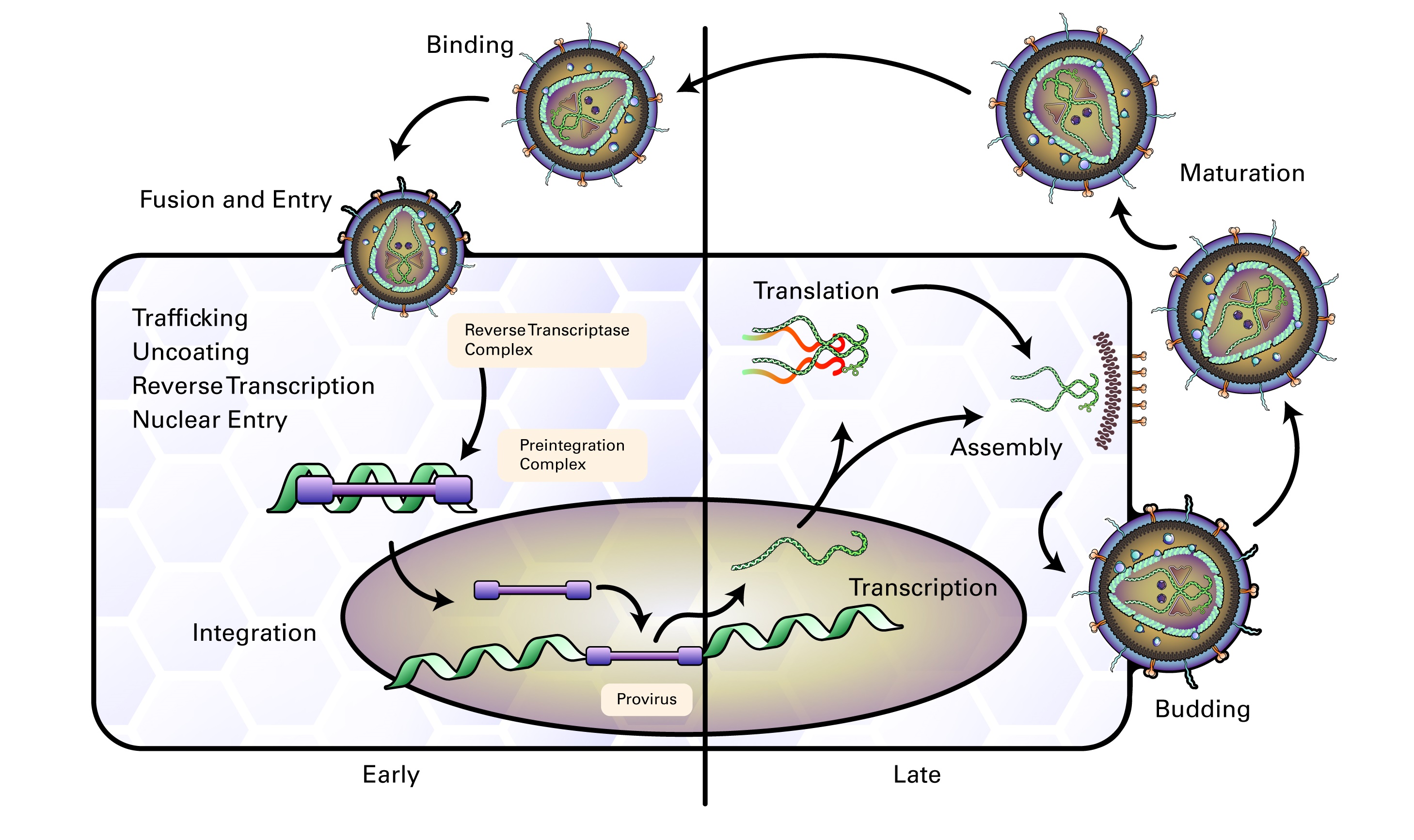
The life cycle of retroviruses can be divided into two phases: the early phase, which includes cell binding through integration of viral DNA into the cell genome, and the late phase, which includes expression of viral genes through the release of virions. Retroviral infection begins when the mature virion envelope glycoprotein interacts with co-receptors on the surface of a cell and fuses membranes of the host cell and virion (Wilen 2011). The viral reverse transcriptase (RT) then uses a host tRNA whose 3’ end is complementary to the sequence near the 5’ end of the viral RNA and acts as a binding site for DNA synthesis. The RT thereby creates an RNA-DNA duplex, which is a substrate for RNase H to remove the 5’ end of the viral RNA, resulting in a newly synthesized minus-strand DNA which can then act, through a complex series of steps, as a template for synthesizing linear double stranded DNA (reviewed by Hu, 2012). The double stranded DNA template is circularized and transported to the nucleus, where it integrates into the host genome, is then transcribed back into viral RNA which is translated to produce viral proteins, and the capsids are then assembled into viral particles and budded from the cell (Nisole, S 2004).
The Delta retroviruses (HTLV-1 and HTLV-2) have a world-wide distribution, and are commonly found in southwest Japan, the Caribbean, and West Africa, where the incidence rate can be 30% of the population. The virus is spread through sexual intercourse, blood transfusions, and by breast feeding. Infection is mostly asymptomatic, however, a subset of infected individuals can develop one of the following diseases: T-cell leukemia/lymphoma, an aggressive tumor of CD4 cells that occurs in 5% of infected individuals, HTLV-1 associated myelopathy (tropical spastic paraparesis), a non-demyelinating disorder with weakness in the lower limbs, infective dermatitis, or uveitis.
The Lentiviruses (HIV-1 and HIV-2) are the causative agent of acquired immune deficiency syndrome (AIDS). AIDS is a new disease that arose in 1980, and HIV-1 is highly homologous to the simian immunodeficiency viruses that infect African monkeys and have crossed the species barrier. HIV-2 is most homologous to an SIV strain that entered the human population in the 1940s. Between humans, both viruses are most commonly transmitted through sexual intercourse, but can also be transmitted vertically in utero, at childbirth, or through breast feeding.
The clinical course of HIV begins with a self-limiting febrile infection 2-4 weeks after exposure with symptoms of fever, lymphadenopathy, and diarrhea, during which time the host develops antibodies. This phase is followed by an asymptomatic phase that can last years, when the patient enters clinical latency. They are infectious but largely asymptomatic, and also test positive for HIV antibodies.
The prodromal phase begins with a drop in the CD4+ count, when patients begin to experience weight loss, fever, lymphadenopathy, oral candidiasis, and diarrhea. The disease then progresses to AIDS, which is characterized by fever, diarrhea, weight loss, skin rashes, neuropathy, myelopathy, dementia, and increased susceptibility to opportunistic infections such as TB, HSV, oral candidiasis, cryptosporidiosis, pneumocystis infections, other viral and fungal infections, and virally induced malignancies such as Kaposi’s sarcoma, hairy leukoplakia, and lymphomas.
The disease pathogenesis begins when HIV infects dendritic antigen processing cells (DC) in the oral and genital mucosa, attaching to these cells via a DC SIGN receptor on the cell surface. HIV particles are then transported to CD4+ helper T-cells where many cycles of infection are established in lymphoid tissues. Loss of function and death of CD4+ helper T-cells results in a drop in the helper T-cell count. Clinical disease progression is correlated with the loss of CD4+ cells and may take five to ten years to progress from primary infection to complete failure of the T-cell mediated immune response.
Early diagnosis is dependent upon the detection of viral DNA or RNA by PCR, or by the detection of p24 antigen in serum. The p24 antigen is usually cleared once patient antibodies appear, so this test is most useful during the early window period, which occurs six days before the development of antibodies and 3-5 weeks after initial exposure.
Treatment of HIV infection includes antiviral agents that disrupt the viral replication cycle. A regimen of three drugs (HAART) given simultaneously suppresses HIV replication, but these drugs are not cures; they must be taken for life to maintain viral suppression. These drugs include nucleoside and nucleotide reverse transcriptase inhibitors, non-nucleoside transcriptase inhibitors, and protease inhibitors. Short courses of anti-retroviral drugs have also been effective in preventing HIV infection following exposure.
There is no currently effective vaccine for the prevention of HIV infection, largely due to:
- A) Variability of the envelope antigens.
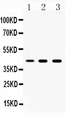
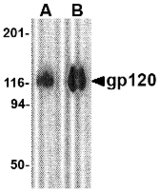
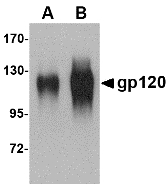
- B) Heavily glycosylated or hidden nature of epitopes of the envelope glycoprotein gp120.
- C) Presence of specific antibodies have been shown to actually enhance infection because antibody coated viruses can bind to Fc receptors on the surface of cells.
References
- Hu, WS and Hughes, SH. HIV-1 Reverse Transcription (2012). Cold Spring Harbor Perspect Med Vol 2 (10):a006882. doi: 10.1101/cshperspect.a006882
- Katzourakis, A. Origin of the retroviruses: when, where and how? (2017) Curr Opin Virol 25:23-27. doi: 10.1016/j.coviro.2017.06.006
- Nisole, S and Saib, A. Early steps of the retrovirus replicative cycle. (2001) Retrovirology V1:9. doi: 10.1186/1742-4690-1-9
- Varmus, H. Retroviruses. (1988) Science 240 (4858):1427-35. doi: 10.1126/science.3287617
- Wilen CB, Tilton JC, Doms RW 2011. HIV: Cell binding and entry. Cold Spring Harb Perspect Med 10.1101/cshperspect.a006866 [PMC free article] [PubMed] [CrossRef] [Google Scholar]
All Retroviridae (HIV) Products