order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Locations
Orders Processing,
Shipping & Receiving,
Warehouse
2 Shaker Rd Suites
B001/B101
Shirley, MA 01464
Production Lab
Floor 6, Suite 620
20700 44th Avenue W
Lynnwood, WA 98036
Telephone Numbers
Tel: +1 (206) 374-1102
Fax: +1 (206) 577-4565
Contact Us
Additional Contact Details
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Coronavirus Detection
LSBio’s library of coronavirus products includes numerous kits for the detection of SARS, SARS-CoV-2, MERS, HCoV, FIPV, and other common coronaviruses. We offer COVID-19 real-time quantitative PCR (RT-qPCR) kits for the direct detection of SARS-CoV-2 RNA and a wide range of ELISA kits to detect human IgG/IgM, coronavirus proteins, and host proteins relevant to COVID-19 disease progression.

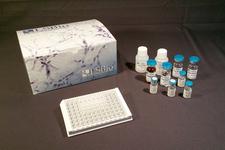
Viral RNA Detection Kits (RT-qPCR)
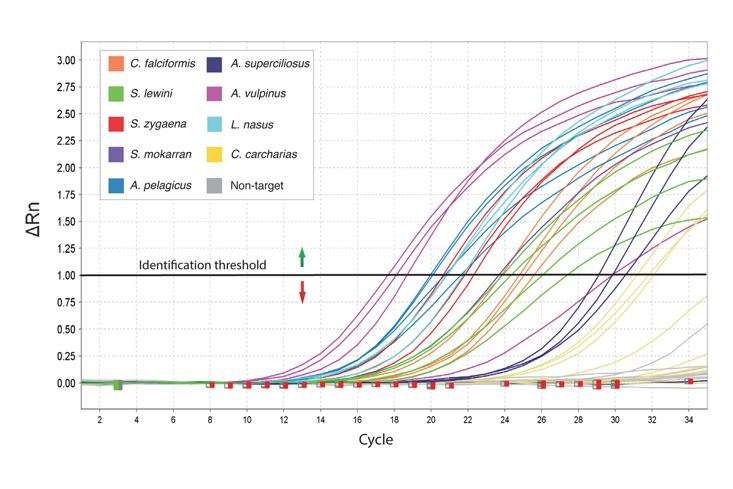
Real-Time qPCR (RT-qPCR) is a technique that allows researchers to quantitatively detect the presence of specific RNA, such as SARS-CoV-2, within a sample. Following nucleic acid extraction and reverse transcription of RNA to cDNA, the polymerase chain reaction is used to amplify the target DNA, which is then detected using fluorescently labeled highly-specific probes. As the reaction progresses, the release of fluorescence can be monitored, providing a means of quantifying the target. In response to the COVID-19 pandemic, we offer both Real-Time qPCR kits and sample collection kits. The RT-qPCR kits require access to a real-time quantitative PCR instrument.
This rapid test is for research use only and has not been approved by the FDA for clinical diagnostic use. Samples collected and processed for this test are potentially infectious, and should be handled only by trained laboratory professionals in a BL-2 laboratory or equivalent setting with a laminar flow hood and personal protective equipment.

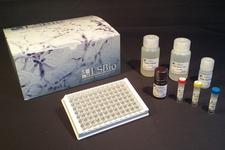
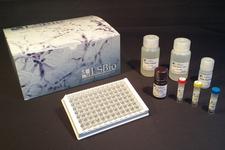
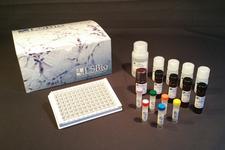
ELISA Detection Kits
LSBio provides a range of Sandwich ELISA kits for the quantitative detection of numerous coronavirus proteins, including SARS-CoV and SARS-CoV-2 spike and nucleocapsid proteins. We also offer kits which detect the SARS-CoV-2 receptor ACE2, protease TMPRSS2, and numerous inflammatory proteins, such as IL-6, IL-10, and TNF-alpha.
All Coronavirus Detection Products