order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Locations
Orders Processing,
Shipping & Receiving,
Warehouse
2 Shaker Rd Suites
B001/B101
Shirley, MA 01464
Production Lab
Floor 6, Suite 620
20700 44th Avenue W
Lynnwood, WA 98036
Telephone Numbers
Tel: +1 (206) 374-1102
Fax: +1 (206) 577-4565
Contact Us
Additional Contact Details
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
order histories, retained contact details for faster checkout, review submissions, and special promotions.
We could not find product '(ID:42472)'. Here are some alternate products to this target that might be of interest to you.
NOX4
NADPH oxidase 4
Constitutive NADPH oxidase which generates superoxide intracellularly upon formation of a complex with CYBA/p22phox. Regulates signaling cascades probably through phosphatases inhibition. May function as an oxygen sensor regulating the KCNK3/TASK-1 potassium channel and HIF1A activity. May regulate insulin signaling cascade. May play a role in apoptosis, bone resorption and lipolysaccharide-mediated activation of NFKB. May produce superoxide in the nucleus and play a role in regulating gene expression upon cell stimulation. Isoform 3 is not functional. Isoform 5 and isoform 6 display reduced activity.Isoform 4: Involved in redox signaling in vascular cells. Constitutively and NADPH-dependently generates reactive oxygen species (ROS). Modulates the nuclear activation of ERK1/2 and the ELK1 transcription factor, and is capable of inducing nuclear DNA damage. Displays an increased activity relative to isoform 1.
| Gene Name: | NADPH oxidase 4 |
| Synonyms: | NOX4, Kidney oxidase-1, KOX, RENOX, KOX-1, NADPH oxidase 4, Renal NAD(P)H-oxidase |
| Target Sequences: | NM_016931 NP_058627.1 Q9NPH5 |
Publications (13)
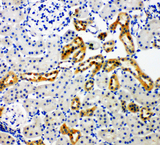
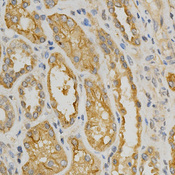

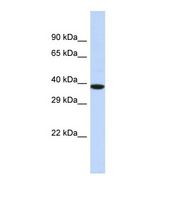

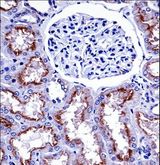
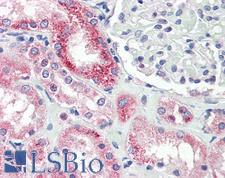
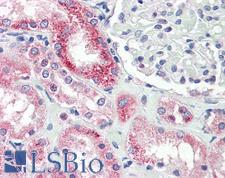




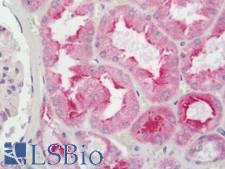
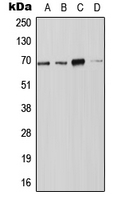
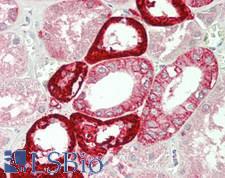

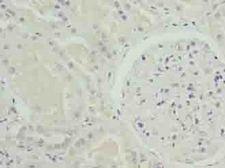
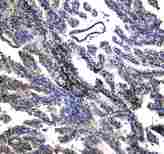
If you do not find the reagent or information you require, please contact Customer.Support@LSBio.com to inquire about additional products in development.











