Login
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Quick Order
Products
Antibodies
ELISA and Assay Kits
Research Areas
Infectious Disease
Resources
Purchasing
Reference Material
Contact Us
Locations
Orders Processing,
Shipping & Receiving,
Warehouse
2 Shaker Rd Suites
B001/B101
Shirley, MA 01464
Production Lab
Floor 6, Suite 620
20700 44th Avenue W
Lynnwood, WA 98036
Telephone Numbers
Tel: +1 (206) 374-1102
Fax: +1 (206) 577-4565
Contact Us
Additional Contact Details
Login
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Quick Order
| Catalog Number | Size | Price |
|---|---|---|
| LS-C58750-100 | 100 Tests | $485 |
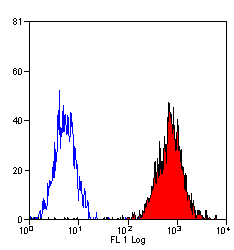
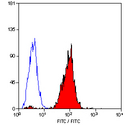
Monoclonal Mouse anti‑Human HLA‑B7 Antibody (clone BB7.1, RPE) LS‑C58750
Monoclonal Mouse anti‑Human HLA‑B7 Antibody (clone BB7.1, RPE) LS‑C58750
Antibody:
HLA-B7 Mouse anti-Human Monoclonal (RPE) (BB7.1) Antibody
Application:
Flo
Reactivity:
Human
Format:
R. Phycoerythrin, Unmodified
Other formats:
Toll Free North America
 206-374-1102
206-374-1102
For Research Use Only
Overview
Antibody:
HLA-B7 Mouse anti-Human Monoclonal (RPE) (BB7.1) Antibody
Application:
Flo
Reactivity:
Human
Format:
R. Phycoerythrin, Unmodified
Other formats:
Specifications
Description
HLA-B7 antibody LS-C58750 is an RPE-conjugated mouse monoclonal antibody to human HLA-B7. Validated for Flow.
Host
Mouse
Reactivity
Human
(tested or 100% immunogen sequence identity)
Clonality
IgG1
Monoclonal
Clone
BB7.1
Conjugations
R. Phycoerythrin.
Also available Unconjugated or conjugated with FITC.
Purification
Affinity purified
Modifications
Unmodified
Immunogen
Papain solubilized HLA-B7 antigen.
Specificity
Binds to the HLA-B7 antigen and does not cross-react with HLA-B27 or other related HLA antigens. It can be used to distinguish true HLA-B27 positives from false HLA-B27 positives (i. e. HLA-B7 positive) in the investigation of such diseases as ankylosing spondylitis and anterior uveitis.
Applications
- Flow Cytometry (1:1)
- Applications tested for the base form of this product only
Usage
The applications listed have been tested for the unconjugated form of this product. Other forms have not been tested. Flow Cytometry: Use 10 ul of the suggested working dilution to label 1 x 10^6 cells or 100 ul whole blood. Method sheets are available on request.
Presentation
PBS, 0.09% Sodium Azide, 5% Sucrose, 1% BSA
Reconstitution
Reconstitute with 1.0 ml distilled H2O.
Storage
Store at 4°C. Do not freeze. Store undiluted. Protect from light. Microcentrifugation recommended if solution contains precipitate.
Restrictions
For research use only. Intended for use by laboratory professionals.
Publications (0)
Customer Reviews (0)
Featured Products
Species:
Human, Non-Human Primates
Applications:
Flow Cytometry
Species:
Human, Mouse, Rat
Applications:
IHC, Western blot, Immunoprecipitation, ELISA
Species:
Human, Mouse, Rat
Applications:
IHC, IHC - Paraffin, Western blot, Peptide Enzyme-Linked Immunosorbent Assay
Request SDS/MSDS
To request an SDS/MSDS form for this product, please contact our Technical Support department at:
Technical.Support@LSBio.com
Requested From: United States
Date Requested: 4/26/2024
Date Requested: 4/26/2024