order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Locations
Orders Processing,
Shipping & Receiving,
Warehouse
2 Shaker Rd Suites
B001/B101
Shirley, MA 01464
Production Lab
Floor 6, Suite 620
20700 44th Avenue W
Lynnwood, WA 98036
Telephone Numbers
Tel: +1 (206) 374-1102
Fax: +1 (206) 577-4565
Contact Us
Additional Contact Details
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Immunohistochemistry Protocol for Double-Labeling of Formalin-Fixed, Paraffin Embedded Tissues (For Visualizing Two Different Primary Antibodies (DAB and Vector Red)
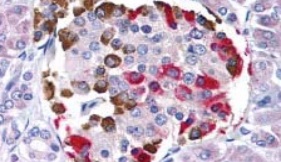
Co-localization IHC experiments are performed to determine the distribution of staining of two different antibodies on the same tissue section. This method allows the investigator to identify whether the same cell is expressing both antigens, and where they are localizing within the cell. The procedure is performed sequentially, by first performing immunochemistry with an antibody using an AP-Vector Red detection system, followed by applying the second antibody using an HRP-DAB detection system. The resulting colorometric precipitates allow for simultaneous detection of both targets on a single section because the first antibody will produce a red deposit, and the second will appear brown. When the antibody co-localizes, the deposit will appear reddish-brown. Prior to performing co-localization IHC, it is important to test each primary antibody individually and determine the antibody staining concentration range that produces the optimal signal for that tissue sample.
Tissue Sectioning, Deparaffinization, and Rehydration
- Section paraffin blocks into 4 micron sections with microtome and place on charged microscope slides (Fisher, ProbeOn, Cat. #22230900).
- Heat slides in a tissue-drying oven for 45 minutes at 60°C.
- Wash slides in 3 changes of xylene for 5 minutes each at room temperature.
- Wash slides in 3 changes of 100% alcohol for 3 minutes each at room temperature.
- Wash slides in 2 changes of 95% alcohol for 3 minutes each at room temperature.
- Wash slides in 1 change of 80% alcohol for 3 minutes at room temperature.
- Rinse slides in running distilled water for 5 minutes at room temperature.
Antigen Retrieval
- Steam slides in 0.01 M sodium citrate buffer, pH 6.0, for 20 minutes at 99-100°C.
- Remove slides from heat and hold in citrate buffer for 20 minutes at room temperature.
- Rinse slides in 1X TBS with Tween (TBST) for 1 minute at room temperature.
The following steps are to be conducted at room temperature. Do not allow tissues to dry at any time during the staining procedure.
Immunostaining
Application of Primary Antibody to First Target:
- Apply peroxidase block (3% hydrogen peroxide) to the slides and incubate for 5 minutes.
- Rinse slides in 1X TBST for 1 minute.
- Apply Universal Protein Block (DAKO, Cat. #X0909) to the slides and incubate for 20 minutes.
- Drain protein block from slides.
- Apply the primary antibody to the first target (to be visualized with brown DAB chromogen) to the slides and incubate for 45 minutes.
- Rinse slides in 1X TBST for 1 minute.
- Use 1 of the following 3 methods for the detection of the primary antibody to first target:
- Apply a biotinylated anti-IgG (H+L) secondary antibody to the slides and incubate for 30 minutes.
- Rinse slides in 1X TBST for 1 minute.
- Apply HRP-streptavidin (Vector PK4000) to the slides and incubate for 30 minutes.
- Rinse slides in 1X TBST for 1 minute.
- Apply LSAB2 System-HRP LINK solution (DAKO, Cat. #K0679) to the slides and incubate for 15 minutes.
- Rinse slides in 1X TBST for 1 minute.
- Apply LSAB2 System-HRP Streptavidin-HRP solution (DAKO, Cat. #K0679) to the slides and incubate for 10 minutes.
- Rinse slides in 1X TBST for 1 minute.
- Apply DAKO Envision+ HRP labeled polymer (Cat. #K4001 mouse primary; Cat. #K4003 rabbit primary) to the slides and incubate for 30 minutes.
- Rinse slides in 1X TBST for 1 minute.
- Apply prepared DAB Substrate-Chromogen solution (DAKO, Cat. #K3468) to the slides and incubate for 5 minutes.
- Rinse slides in 1X TBST for 1 minute.
Vector HRP System:
Dako LSAB2-HRP System:
DAKO Envision+ HRP/DAB System:
Application of Primary Antibody for Second Target:
- Apply peroxidase block (3% hydrogen peroxide) to the slides and incubate for 5 minutes.
- Rinse slides in 1X TBST for 1 minute.
- Apply a Universal Protein Block (DAKO, Cat. #X0909) to the slides and incubate for 20 minutes.
- Drain protein block from slides.
- Apply the primary antibody to the second target (to be visualized with fuchsia Vector Red chromogen) to the slides and incubate for 45 minutes.
- Rinse slides in 1X TBST for 1 minute.
- Apply a biotinylated anti-host (H+L) secondary antibody to the slides and incubate for 30 minutes.
- Rinse slides in 1X TBST for 1 minute.
- Apply alkaline phosphatase streptavidin (Vector, Cat. #AK-5000) to the slides and incubate for 30 minutes.
- Rinse slides in 1X TBST for 1 minute.
- Apply alkaline phosphatase chromogen substrate (Vector, Cat. #AK-5100) to the slides and incubate for 30 minutes.
- Rinse slides in 1X TBST for 1 minute.
Counterstaining with Hematoxylin
- Stain slides with 65% Harris' Hematoxylin for 1 minute. Hematoxylin stains nucleic acids (nuclei) a deep blue-purple.
Dehydration and Coverslipping
- This method should only be used if the chromogen substrate used is alcohol insoluble (e.g. Vector Red or DAB).
- Wash slides in 2 changes of 80% alcohol for 1 minute each.
- Wash slides in 2 changes of 95% alcohol for 1 minute each.
- Wash slides in 3 changes of 100% alcohol for 1 minute each.
- Wash slides in 3 changes of xylene for 1 minute each.
- Apply coverslip with a drop of permanent mounting medium.
Sample Double-Label Immunohistochemistry Report
For more information about LSBio Protocols contact Technical.Support@LSBio.com