Login
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Quick Order
Products
Antibodies
ELISA and Assay Kits
Research Areas
Infectious Disease
Resources
Purchasing
Reference Material
Contact Us
Locations
Orders Processing,
Shipping & Receiving,
Warehouse
2 Shaker Rd Suites
B001/B101
Shirley, MA 01464
Production Lab
Floor 6, Suite 620
20700 44th Avenue W
Lynnwood, WA 98036
Telephone Numbers
Tel: +1 (206) 374-1102
Fax: +1 (206) 577-4565
Contact Us
Additional Contact Details
Login
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Quick Order
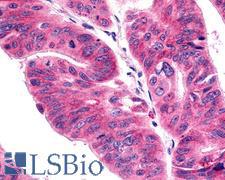
PathPlusTM ACKR4 / CCRL1 / CCR11 Antibodies
Atypical chemokine receptor 4 (ACKR4) controls chemokine levels and localization via high-affinity chemokine binding that is uncoupled from classic ligand-driven signal transduction cascades, resulting instead in chemokine sequestration, degradation, or transcytosis. Chemokine-binding does not activate G-protein-mediated signal transduction but instead induces beta-arrestin recruitment, leading to ligand internalization. ACKR4 plays an important role in controlling the migration of immune and cancer cells that express chemokine receptors CCR7 and CCR9, by reducing the availability of CCL19, CCL21, and CCL25 through internalization. ACKR4 negatively regulates CXCR3-induced chemotaxis and regulates T-cell development in the thymus.
References: The UniProt Consortium. Nucleic Acids Res. 47: D506-515 (2019); Nucleic Acids Res. 2016 Jan 4;44(D1):D733-45, PMID:26553804
1 PathPlusTM Antibody
☰ Filters
Products
Antibodies
(1)
Type
Primary
(1)
Target
ACKR4 / CCRL1 / CCR11
(1)
Reactivity
Human
(1)
Mouse
(1)
Rat
(1)
Bat
(1)
Dog
(1)
Horse
(1)
Pig
(1)
Rabbit
(1)
Application
IHC
(1)
IHC-P
(1)
Host
rabbit
(1)
Product Group
GPCR Database Antibodies
(1)
PathPlus Neuro
(1)
Clonality
polyclonal pc
(1)
Format
Unconjugated
(1)
Epitope
Cytoplasmic Domain
(1)
Publications
No
(1)

Neuroscience
Fast Shipping
ACKR4 / CCRL1 / CCR11 Rabbit anti-Human Polyclonal (Cytoplasmic Domain) Antibody
Rabbit, Mouse, Dog, Rat, Pig, Horse, Bat, Human
IHC, IHC-P
Unconjugated
50 µg/$440
Viewing 1-1
of 1
product results










