Login
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Quick Order
Products
Antibodies
ELISA and Assay Kits
Research Areas
Infectious Disease
Resources
Purchasing
Reference Material
Contact Us
Locations
Orders Processing,
Shipping & Receiving,
Warehouse
2 Shaker Rd Suites
B001/B101
Shirley, MA 01464
Production Lab
Floor 6, Suite 620
20700 44th Avenue W
Lynnwood, WA 98036
Telephone Numbers
Tel: +1 (206) 374-1102
Fax: +1 (206) 577-4565
Contact Us
Additional Contact Details
Login
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Quick Order
| Catalog Number | Size | Price |
|---|---|---|
| LS-B11760-50 | 50 µg (0.5 mg/ml) | $485 |





1 of 3
2 of 3
3 of 3
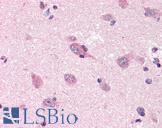
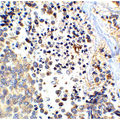
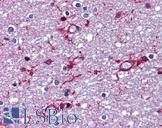
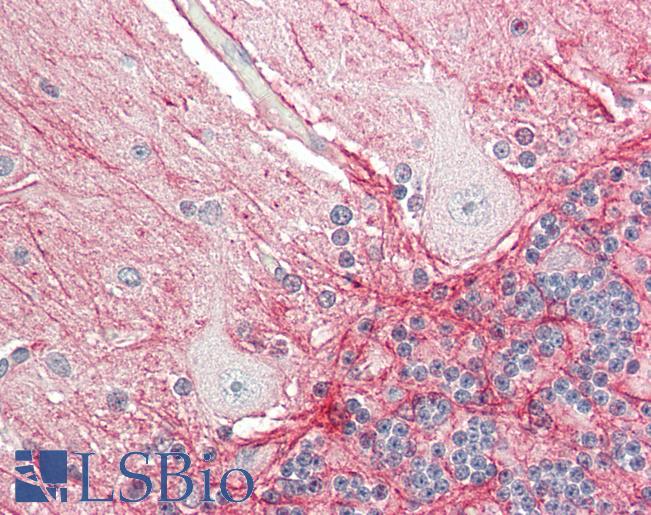
PathPlus™ Polyclonal Goat anti‑Mouse APP / Beta Amyloid Precursor Antibody (N‑Terminus, IHC, WB) LS‑B11760
PathPlus™ Polyclonal Goat anti‑Mouse APP / Beta Amyloid Precursor Antibody (N‑Terminus, IHC, WB) LS‑B11760
Note: This antibody replaces LS-C154911, LS-C193925
Antibody:
APP / Beta Amyloid Precursor Goat anti-Mouse Polyclonal (N-Terminus) Antibody
Application:
IHC, IHC-P, WB, Peptide-ELISA
Reactivity:
Mouse, Human, Rat
Format:
Unconjugated, Unmodified
Toll Free North America
 206-374-1102
206-374-1102
For Research Use Only
Overview
Antibody:
APP / Beta Amyloid Precursor Goat anti-Mouse Polyclonal (N-Terminus) Antibody
Application:
IHC, IHC-P, WB, Peptide-ELISA
Reactivity:
Mouse, Human, Rat
Format:
Unconjugated, Unmodified
Specifications
Description
APP (Beta Amyloid Precursor) is a protein that functions as a cell surface receptor and performs physiological functions on the surface of neurons relevant to neurite growth, neuronal adhesion and axonogenesis. It is involved in cell mobility and transcription regulation through protein-protein interactions and can promote transcription activation through binding to APBB1-KAT5 and inhibits Notch signaling through interaction with Numb. It couples to apoptosis-inducing pathways such as those mediated by G(O) and JIP and also inhibits G(o) alpha ATPase activity. It functions as a kinesin I membrane receptor, mediating the axonal transport of beta-secretase and presenilin 1. It is also involved in copper homeostasis/oxidative stress through copper ion reduction. In vitro, copper-metallated APP induces neuronal death directly or is potentiated through Cu2+-mediated low-density lipoprotein oxidation. It can regulate neurite outgrowth through binding to components of the extracellular matrix such as heparin and collagen I and IV. APP induces a AGER-dependent pathway that involves activation of p38 MAPK, resulting in internalization of amyloid-beta peptide and leading to mitochondrial dysfunction in cultured cortical neurons.
References: The UniProt Consortium. Nucleic Acids Res. 47: D506-515 (2019); Nucleic Acids Res. 2016 Jan 4;44(D1):D733-45, PMID:26553804
Target
Mouse APP / Beta Amyloid Precursor
Synonyms
APP | ABPP | A4 | ABETA | AD1 | Alzheimer disease | Amyloid beta | Beta-amyloid peptide | APPI | Beta amyloid | CVAP | AAA | PN2 | PreA4 | Amyloid beta A4 protein | Peptidase nexin-II | CTFgamma | PN-II | Protease nexin-II
Host
Goat
Reactivity
Mouse, Human, Rat
(tested or 100% immunogen sequence identity)
Clonality
Polyclonal
Conjugations
Unconjugated
Purification
Purified from goat serum by ammonium sulphate precipitation followed by antigen affinity chromatography using the immunizing peptide.
Modifications
Unmodified
Immunogen
Peptide with sequence DAEFGHDSGFEVRHQK-C, from the N-Terminus of protein sequence according to NP_001185752.1NP_031497.2NP_001185753.1NP_001185754.1NP_001185755.1.
Epitope
N-Terminus
Specificity
Mouse APP. This antibody is expected to recognize all reported isoforms of mAPP (NP_001185752.1; NP_031497.2; NP_001185753.1; NP_001185754.1; NP_001185755.1) and to recognize all lengths of the putative mouse Amyloid beta peptide.
Applications
- IHC
- IHC - Paraffin (5 µg/ml)
- Western blot (0.03 - 0.1 µg/ml)
- Peptide Enzyme-Linked Immunosorbent Assay (1:64000)
|
Performing IHC? See our complete line of Immunohistochemistry Reagents including antigen retrieval solutions, blocking agents
ABC Detection Kits and polymers, biotinylated secondary antibodies, substrates and more.
|
Usage
Peptide ELISA: antibody detection limit dilution 1:64000. Western blot: Approx 100kD band observed in Mouse and Rat Brain lysates (calculated MW of 86.7kD according to NP_001185752.1). Recommended concentration: 0.03-0.1 ug/ml.
Presentation
TBS, pH 7.3, 0.02% Sodium Azide, 0.5% BSA
Storage
Aliquot and store at -20°C. Avoid freeze-thaw cycles.
Restrictions
For research use only. Intended for use by laboratory professionals.
About APP / Beta Amyloid Precursor
Validation
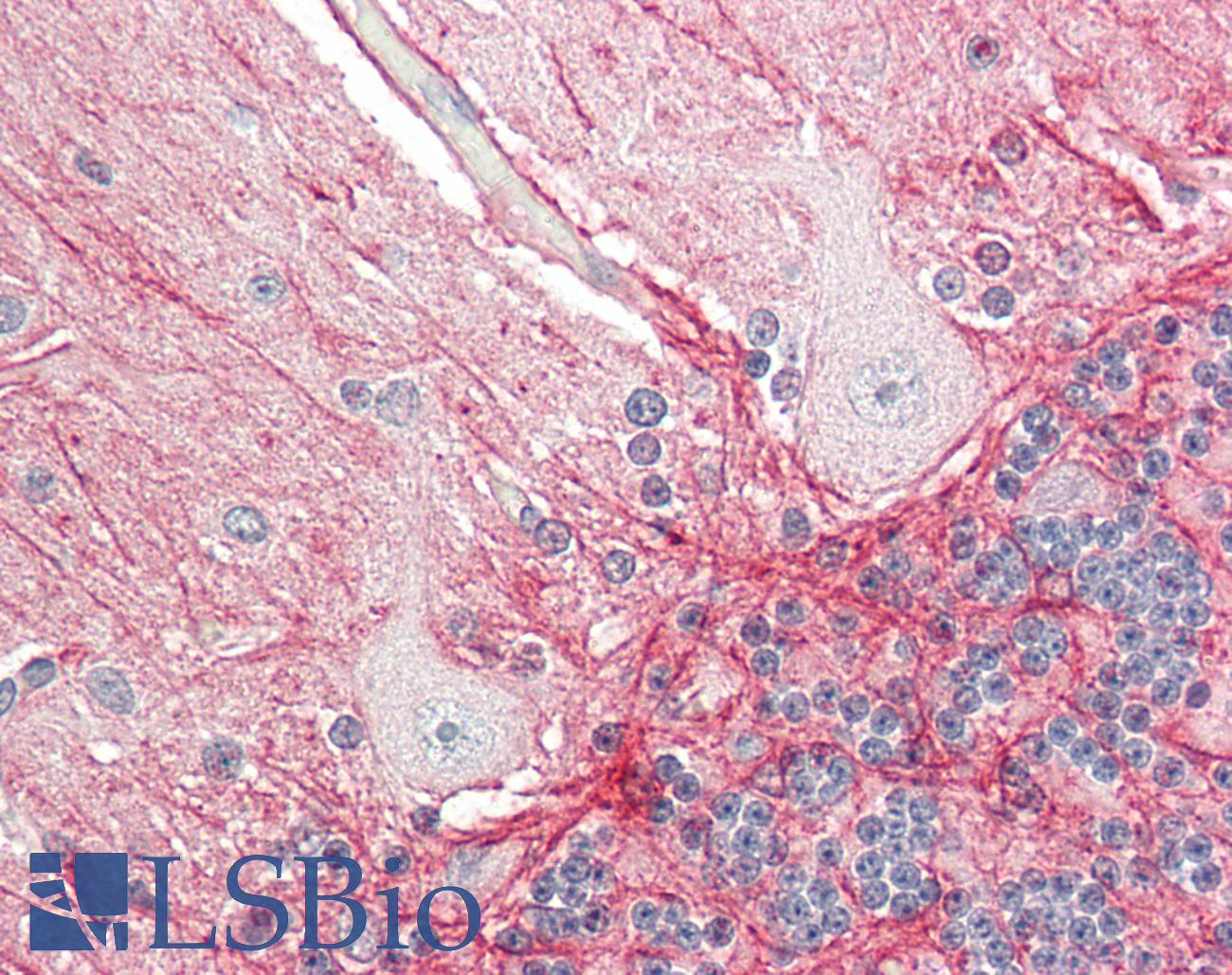
Human Brain, Cerebellum: Formalin-Fixed, Paraffin-Embedded (FFPE)
Human Brain, Cerebellum: Formalin-Fixed, Paraffin-Embedded (FFPE)
See More About...
LSBio Ratings
PathPlus™ APP / Beta Amyloid Precursor Antibody (N-Terminus) for IHC, WB/Western LS-B11760 has an LSBio Rating of
Laboratory Validation Score (5)
Learn more about The LSBio Ratings Algorithm
Publications (0)
Customer Reviews (0)
Featured Products
Species:
Human, Mouse
Applications:
IHC, IHC - Paraffin, Western blot, ELISA
Species:
Human, Mouse, Rat
Applications:
IHC, Western blot, ELISA
Species:
Human, Mouse, Rat
Applications:
IHC, Western blot
Species:
Human, Mouse, Rat
Applications:
IHC, IHC - Paraffin, Immunofluorescence, Western blot, ELISA
Species:
Human, Mouse, Rat, Primate
Applications:
Western blot, ELISA
Reactivity:
Rat
Range:
1.56-100 ng/ml
Request SDS/MSDS
To request an SDS/MSDS form for this product, please contact our Technical Support department at:
Technical.Support@LSBio.com
Requested From: United States
Date Requested: 7/26/2024
Date Requested: 7/26/2024