Login
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Quick Order
Products
Antibodies
ELISA and Assay Kits
Research Areas
Infectious Disease
Resources
Purchasing
Reference Material
Contact Us
Locations
Orders Processing,
Shipping & Receiving,
Warehouse
2 Shaker Rd Suites
B001/B101
Shirley, MA 01464
Production Lab
Floor 6, Suite 620
20700 44th Avenue W
Lynnwood, WA 98036
Telephone Numbers
Tel: +1 (206) 374-1102
Fax: +1 (206) 577-4565
Contact Us
Additional Contact Details
Login
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Quick Order
Vaccinia Virus
Vaccinia Virus Target Details
| Target Name: | Vaccinia Virus |
☰ Filters
Products
Antibodies
(12)
Proteins
(1)
Type
Primary
(12)
Recombinant
(1)
Target
Vaccinia Virus
(13)
Reactivity
Vaccinia Virus
(10)
Virus
(2)
Application
IHC
(5)
IHC-Fr
(1)
WB
(2)
ELISA
(12)
IF
(8)
Host
rabbit
(7)
mouse
(5)
Isotype
IgG
(1)
IgG1
(2)
IgG2a
(1)
IgG2b
(2)
Clonality
monoclonal mc
(5)
polyclonal pc
(7)
Clone
TV43
(1)
Format
Biotin Conjugated
(1)
FITC Conjugated
(1)
HRP Conjugated
(2)
Unconjugated
(8)
Publications
No
(13)
Tag
His,C-terminus
(1)
Species
Vaccinia Virus
(1)
Source
Baculovirus
(1)
Low Endotoxin Level
Low endotoxin level
(1)
Vaccinia Virus Rabbit anti-Vaccinia Virus Polyclonal Antibody
Vaccinia Virus
ELISA, IF, IHC
Unconjugated
100 µg/$387
Vaccinia Virus Rabbit anti-Vaccinia Virus Polyclonal (Biotin) Antibody
Vaccinia Virus
ELISA, IF, IHC
Biotin Conjugated
100 µg/$398
Vaccinia Virus Rabbit anti-Vaccinia Virus Polyclonal (FITC) Antibody
Vaccinia Virus
ELISA, IF, IHC
FITC Conjugated
100 µg/$398
Vaccinia Virus Rabbit anti-Vaccinia Virus Polyclonal (HRP) Antibody
Vaccinia Virus
ELISA, IHC
HRP Conjugated
1 ml/$785
Vaccinia Virus Mouse anti-Vaccinia Virus Monoclonal Antibody
Vaccinia Virus
ELISA
Unconjugated
100 µg/$413
Vaccinia Virus Mouse anti-Vaccinia Virus Monoclonal Antibody
Vaccinia Virus
ELISA, IF
Unconjugated
100 µg/$413
Vaccinia Virus Rabbit anti-Vaccinia Virus Polyclonal Antibody
Vaccinia Virus
ELISA, IF, WB
Unconjugated
500 µl/$355
Vaccinia Virus Rabbit anti-Vaccinia Virus Polyclonal (HRP) Antibody
Vaccinia Virus
ELISA, IF, IHC
HRP Conjugated
100 µg/$413
Vaccinia Virus Mouse anti-Vaccinia Virus Monoclonal Antibody
Vaccinia Virus
ELISA, IF
Unconjugated
100 µg/$413
Vaccinia Virus Mouse anti-Vaccinia Virus Monoclonal Antibody
Vaccinia Virus
ELISA, IF
Unconjugated
100 µg/$413
Vaccinia Virus Mouse anti-Virus Monoclonal (TV43) Antibody
Virus
ELISA, WB
Unconjugated
0.2 mg/$460
Vaccinia Virus Rabbit anti-Virus Polyclonal Antibody
Virus
ELISA, IHC-Fr
Unconjugated
1 ml/$518
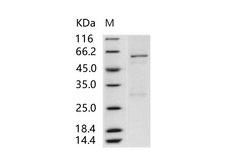
Baculovirus
His,C-terminus
Less than 1.0 EU/µg protein (determined by LAL method).
39.8KDa
100 µg/$843
Viewing 1-13
of 13
product results
If you do not find the reagent or information you require, please contact Customer.Support@LSBio.com to inquire about additional products in development.









