order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Locations
Orders Processing,
Shipping & Receiving,
Warehouse
2 Shaker Rd Suites
B001/B101
Shirley, MA 01464
Production Lab
Floor 6, Suite 620
20700 44th Avenue W
Lynnwood, WA 98036
Telephone Numbers
Tel: +1 (206) 374-1102
Fax: +1 (206) 577-4565
Contact Us
Additional Contact Details
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
order histories, retained contact details for faster checkout, review submissions, and special promotions.
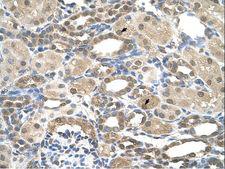
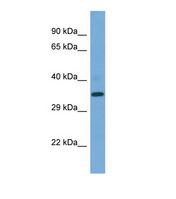
PSEN2 / Presenilin 2
presenilin 2 (Alzheimer disease 4)
Alzheimer's disease (AD) patients with an inherited form of the disease carry mutations in the presenilin proteins (PSEN1 or PSEN2) or the amyloid precursor protein (APP). These disease-linked mutations result in increased production of the longer form of amyloid-beta (main component of amyloid deposits found in AD brains). Presenilins are postulated to regulate APP processing through their effects on gamma-secretase, an enzyme that cleaves APP. Also, it is thought that the presenilins are involved in the cleavage of the Notch receptor such that, they either directly regulate gamma-secretase activity, or themselves act are protease enzymes. Two alternatively spliced transcript variants encoding different isoforms of PSEN2 have been identified.
| Gene Name: | presenilin 2 (Alzheimer disease 4) |
| Family/Subfamily: | Protease , Aspartic A22A |
| Synonyms: | PSEN2, AD3LP, AD4, AD5, AD3L, CMD1V, PS-2, PSNL2, PS2, STM2, Presenilin 2, STM-2, E5-1, Presenilin-2 |
| Target Sequences: | NM_000447 NP_000438.2 P49810 |
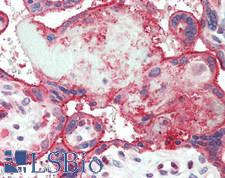
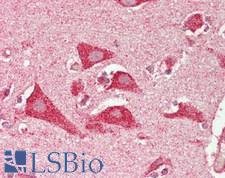
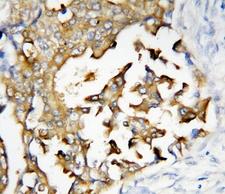
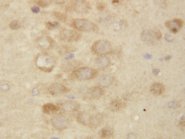
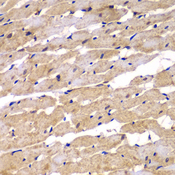
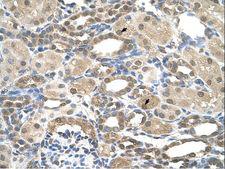
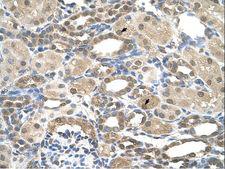
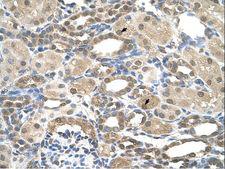



If you do not find the reagent or information you require, please contact Customer.Support@LSBio.com to inquire about additional products in development.










