Login
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Quick Order
Products
Antibodies
ELISA and Assay Kits
Research Areas
Infectious Disease
Resources
Purchasing
Reference Material
Contact Us
Locations
Orders Processing,
Shipping & Receiving,
Warehouse
2 Shaker Rd Suites
B001/B101
Shirley, MA 01464
Production Lab
Floor 6, Suite 620
20700 44th Avenue W
Lynnwood, WA 98036
Telephone Numbers
Tel: +1 (206) 374-1102
Fax: +1 (206) 577-4565
Contact Us
Additional Contact Details
Login
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Quick Order
| Catalog Number | Size | Price |
|---|---|---|
| LS-C59538-1 | 1 mg (1 mg/ml) | $606 |
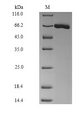
![MBP Tag Antibody - Anti-Maltose Binding Protein (MBP) Epitope Tag Antibody - Western Blot. Anti-MBP epitope tag polyclonal antibody detects MBP-tagged recombinant proteins by western blot. Polyclonal rabbit-anti-MBP epitope tag at 0.5-1.0 ug/ml was used to detect 1.0 ug of recombinant protein containing the MBP epitope tag. The apparent molecular weight of this band is 42 kD. A minor band at corresponding to multimers of this protein is also evident. A 4-20% gradient gel was used to separate the protein by SDS-PAGE. The protein was transferred to nitrocellulose using standard methods. After blocking the membrane was probed with the primary antibody for 1 h at room temperature followed by washes and reaction with a 1:2500 dilution of IRDye 800 conjugated Gt-a-Rabbit IgG [H&L] (code for 30 min at room temperature. LICORs Odyssey Infrared Imaging System was used to scan and process the image. Other detection systems will yield similar results.](https://lsbio-7d62.kxcdn.com/image2/mbp-tag-antibody-ls-c59538/62668_185424.jpg)
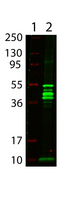
![MBP Tag Antibody - Anti-Maltose Binding Protein (MBP) Epitope Tag Antibody - Western Blot. Anti-MBP epitope tag polyclonal antibody detects MBP-tagged recombinant proteins by western blot. Polyclonal rabbit-anti-MBP epitope tag at 0.5-1.0 ug/ml was used to detect 1.0 ug of recombinant protein containing the MBP epitope tag. The apparent molecular weight of this band is 42 kD. A minor band at corresponding to multimers of this protein is also evident. A 4-20% gradient gel was used to separate the protein by SDS-PAGE. The protein was transferred to nitrocellulose using standard methods. After blocking the membrane was probed with the primary antibody for 1 h at room temperature followed by washes and reaction with a 1:2500 dilution of IRDye 800 conjugated Gt-a-Rabbit IgG [H&L] (code for 30 min at room temperature. LICORs Odyssey Infrared Imaging System was used to scan and process the image. Other detection systems will yield similar results.](https://lsbio-7d62.kxcdn.com/image2/mbp-tag-antibody-ls-c59538/62668_185424.jpg)

1 of 2
2 of 2
Polyclonal Rabbit MBP Tag Antibody (WB) LS‑C59538
Polyclonal Rabbit MBP Tag Antibody (WB) LS‑C59538
Antibody:
MBP Tag Rabbit Polyclonal Antibody
Application:
WB, ELISA
Format:
Unconjugated, Unmodified
Toll Free North America
 206-374-1102
206-374-1102
For Research Use Only
Overview
Antibody:
MBP Tag Rabbit Polyclonal Antibody
Application:
WB, ELISA
Format:
Unconjugated, Unmodified
Specifications
Description
MBP Tag antibody LS-C59538 is an unconjugated rabbit polyclonal antibody to MBP Tag. Validated for ELISA and WB.
Host
Rabbit
Clonality
IgG
Polyclonal
Conjugations
Unconjugated
Purification
Antiserum
Modifications
Unmodified
Immunogen
This antibody was purified from whole rabbit serum prepared by repeated immunizations with the MBP epitope tag recombinant protein.
Specificity
This antibody has reacted with all MBP tagged proteins so far tested. In western blotting of bacterial extracts the antibody does not cross-react with endogenous proteins.
Applications
- Western blot (1:1000 - 1:5000)
- ELISA (1:10000 - 1:50000)
Usage
Anti-MBP is optimally suited for monitoring the expression of MBP tagged fusion proteins. As such, anti- MBP/MBP can be used to identify fusion proteins containing the MBP epitope. The antibody recognizes the MBP epitope tag fused to the amino- or carboxy- termini of targeted proteins. This antibody has been tested by ELISA and western blotting against MBP containing recombinant proteins. Although not tested, this antibody is likely functional for immunoprecipitation and immunocytochemistry, and other immunodetection techniques. Maltose binding protein is a bacterial protein, which is often used in protein expression studies because it creates a stable fusion product that does not appear to interfere with the bioactivity of the protein of interest. It also allows for its easy purification from bacterial extracts under mild conditions. Anti-MBP is a companion to the pMAL protein expression system and can be used for the detection and purification of MBP-fusion proteins expressed in E. coli. By Western blot, a band is seen at ~42 kD representing MBP.
Presentation
Lyophilized from 0.02 M Potassium Phosphate, pH 7.2, 0.15 M NaCl, 0.01% Sodium Azide
Reconstitution
Reconstitute in 1 mL of deionized water.
Storage
Short term: store at 4°C. Long term: store at -20°C. Avoid freeze-thaw cycles.
Restrictions
For research use only. Intended for use by laboratory professionals.
Publications (0)
Customer Reviews (0)
Featured Products
Species:
Human, Rat
Applications:
Immunoprecipitation
Source:
Insect
Tag:
9His, C-terminus
Species:
Human
Applications:
IHC, Western blot, Flow Cytometry, ELISA
Request SDS/MSDS
To request an SDS/MSDS form for this product, please contact our Technical Support department at:
Technical.Support@LSBio.com
Requested From: United States
Date Requested: 9/20/2024
Date Requested: 9/20/2024