order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Locations
Orders Processing,
Shipping & Receiving,
Warehouse
2 Shaker Rd Suites
B001/B101
Shirley, MA 01464
Production Lab
Floor 6, Suite 620
20700 44th Avenue W
Lynnwood, WA 98036
Telephone Numbers
Tel: +1 (206) 374-1102
Fax: +1 (206) 577-4565
Contact Us
Additional Contact Details
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
order histories, retained contact details for faster checkout, review submissions, and special promotions.
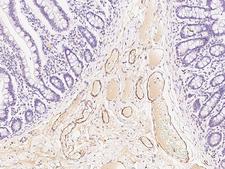
APCS / Serum Amyloid P / SAP
amyloid P component, serum
APCS / Serum Amyloid P / SAP is a glycoprotein, belonging to the pentraxin family of proteins, which has a characteristic pentameric organization. These family members have considerable sequence homology which is thought to be the result of gene duplication. The binding of the encoded protein to proteins in the pathological amyloid cross-beta fold suggests its possible role as a chaperone. This protein is also thought to control the degradation of chromatin. It has been demonstrated that this protein binds to apoptotic cells at an early stage, which raises the possibility that it is involved in dealing with apoptotic cells in vivo.
| Gene Name: | amyloid P component, serum |
| Synonyms: | APCS, 9.5S alpha-1-glycoprotein, Amyloid P component, serum, Pentaxin-related, PTX2, Serum Amyloid P, Serum amyloid P-component, SAP |
| Target Sequences: | NM_001639 NP_001630.1 P02743 |
Publications (1)
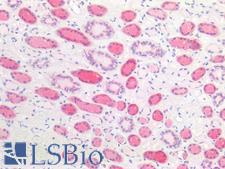
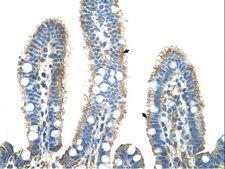
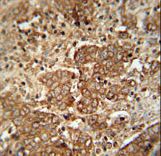


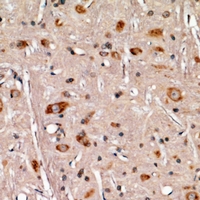

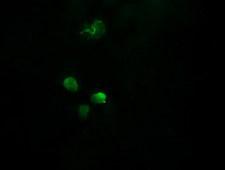

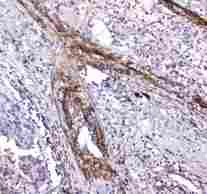

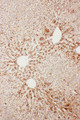
If you do not find the reagent or information you require, please contact Customer.Support@LSBio.com to inquire about additional products in development.










