order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Locations
Orders Processing,
Shipping & Receiving,
Warehouse
2 Shaker Rd Suites
B001/B101
Shirley, MA 01464
Production Lab
Floor 6, Suite 620
20700 44th Avenue W
Lynnwood, WA 98036
Telephone Numbers
Tel: +1 (206) 374-1102
Fax: +1 (206) 577-4565
Contact Us
Additional Contact Details
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
order histories, retained contact details for faster checkout, review submissions, and special promotions.
We could not find product '(ID:109433)'. Here are some alternate products to this target that might be of interest to you.
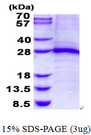
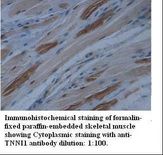
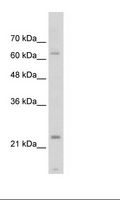
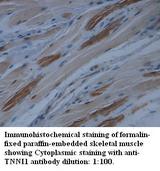
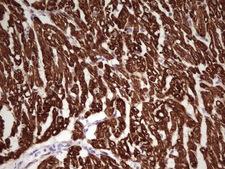
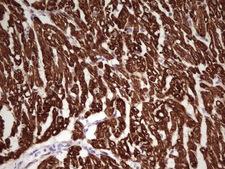
TNNI1
troponin I type 1 (skeletal, slow)
Troponin proteins associate with tropomyosin and regulate the calcium sensitivity of the myofibril contractile apparatus of striated muscles. Troponin I (TnI), along with troponin T (TnT) and troponin C (TnC), is one of 3 subunits that form the troponin complex of the thin filaments of striated muscle. TnI is the inhibitory subunit; blocking actin-myosin interactions and thereby mediating striated muscle relaxation. The TnI subfamily contains three genes: TnI-skeletal-fast-twitch, TnI-skeletal-slow-twitch, and TnI-cardiac. The TnI-fast and TnI-slow genes are expressed in fast-twitch and slow-twitch skeletal muscle fibers, respectively, while the TnI-cardiac gene is expressed exclusively in cardiac muscle tissue. This gene encodes the Troponin-I-skeletal-slow-twitch protein. This gene is expressed in cardiac and skeletal muscle during early development but is restricted to slow-twitch skeletal muscle fibers in adults. The encoded protein prevents muscle contraction by inhibiting calcium-mediated conformational changes in actin-myosin complexes.
| Gene Name: | troponin I type 1 (skeletal, slow) |
| Synonyms: | TNNI1, Troponin I, skeletal, slow, SSTNI, TNN1 |
| Target Sequences: | NM_003281 NP_003272.3 P19237 |
Publications (2)







If you do not find the reagent or information you require, please contact Customer.Support@LSBio.com to inquire about additional products in development.