Login
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Quick Order
Products
Antibodies
ELISA and Assay Kits
Research Areas
Infectious Disease
Resources
Purchasing
Reference Material
Contact Us
Locations
Orders Processing,
Shipping & Receiving,
Warehouse
2 Shaker Rd Suites
B001/B101
Shirley, MA 01464
Production Lab
Floor 6, Suite 620
20700 44th Avenue W
Lynnwood, WA 98036
Telephone Numbers
Tel: +1 (206) 374-1102
Fax: +1 (206) 577-4565
Contact Us
Additional Contact Details
Login
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Quick Order
| Catalog Number | Size | Price |
|---|---|---|
| LS-G3783-10 | 10 µg | $379 |
| LS-G3783-50 | 50 µg | $519 |






1 of 6
2 of 6
3 of 6
4 of 6
5 of 6
6 of 6
Human DLL4 Protein (Recombinant Human IgG1 Fc) (aa1-529) - LS-G3783
Human DLL4 Protein (Recombinant Human IgG1 Fc) (aa1-529) - LS-G3783
Description:
DLL4 Protein LS-G3783 is a Recombinant Human DLL4 produced in HEK 293 Cells aa 1-529 with Human IgG1 Fc tag(s). It is low in endotoxin; Less than 0.01 EU/µg protein (determined by LAL method). For Research Use Only
Toll Free North America
 206-374-1102
206-374-1102
For Research Use Only
Overview
Description:
DLL4 Protein LS-G3783 is a Recombinant Human DLL4 produced in HEK 293 Cells aa 1-529 with Human IgG1 Fc tag(s). It is low in endotoxin; Less than 0.01 EU/µg protein (determined by LAL method). For Research Use Only
Specifications
Type
Recombinant Protein
Target
DLL4
Synonyms
DLL4 | Delta-like 4 protein | Delta like 4 | Delta-like 4 (Drosophila) | Delta-like 4 homolog | Drosophila Delta homolog 4 | Notch ligand DLL4 | Delta 4 | Delta ligand 4 | Delta-like protein 4 | Delta4 | Hdelta2 | Notch ligand delta-2
Species
Human
Modifications
Unmodified
Conjugations
Unconjugated
Tag
Human IgG1 Fc
Region
aa 1-529
Predicted Molecular Weight
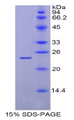
~80kDa (SDS-PAGE)
Expression System
HEK 293 Cells
Source Species
Human
Purification
Greater than 95% by SDS-PAGE
Bio-Activity
Inhibits adipogenesis of 3T3L-1 cells and mesenchymal stem cells (MSCs). Induces Hes-1 in 3T3L-1 cells.
Endotoxin
Less than 0.01 EU/µg protein (determined by LAL method).
Presentation
Lyophilized from PBS, 0.5% Trehalose
Reconstitution
Reconstitute with 50 µl sterile endotoxin-free water.
Storage
Reconstitute, aliquot and store at -20°C (stable for 6 months at this temperature). Avoid freeze-thaw cycles. PBS containing up to 0.1% BSA should be used for further dilutions.
Restrictions
For research use only. Intended for use by laboratory professionals.
About DLL4
Publications (0)
Customer Reviews (0)
Images
Functional Assay
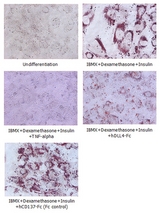
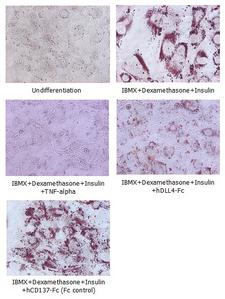
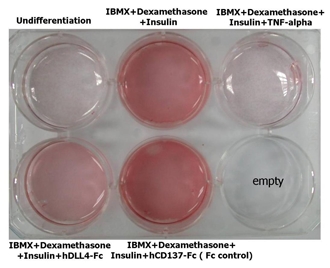
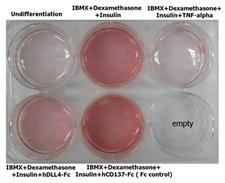
Adipogenesis inhibition of 3T3L-1 cells. 3T3L-1 Cells(mouse pre-adipocyte cells) were maintained in DMEM, supplemented with 10% fetal bovine serum and penicillin-streptomycin. For differentiation of 3T3L-1 Cells, 3T3L-1 Cells were cultured in adipogenic medium which was growth medium supplemented with 1 uM Dexamethasone, 0.5mM IBMX, 10 ug/ml lnsulin (day 0). Medium was changed every 2 days. Staining with Oil Red O was typically performed on day 7. Cells were washed twice with PBS, fixed with 3.7% formalin, and stained with 0.5% filtered Oil Red O in propylene glycol. For negative controls, mouse TNF-alpha (20ng/ml) was added. Recombinant human DLL4-Fc (5 ug/ml) dissolved in DPBS was added to the differentiation medium. These plates were then used to differentiate 3T3L-1 Cells.
Functional Assay
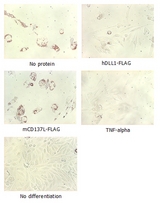
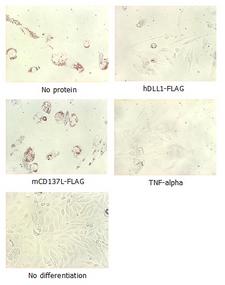
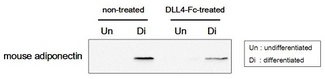
Adipogenesis inhibition of MSCs. MSCs (Mesenchymal stem cells) were maintained in DMEM, supplemented with 10% fetal bovine serum, penicilin-streptomycin and glutamine. For differentiation of MSCs, MSCs were cultured in adipogenic medium which was growth medium supplemented with 1 uM Dexamethasone, 0.5mM IBMX, 10 ug/m lnsulin, 100 uM Indomethacin (day 1). Medium was changed every 3 days. Staining with Oil Red O was typically performed on day 30. For negative controls, TNF-alpha (20ng/ml) was added. To immobilize Notch ligands on the plastic surface of the culture plates, plates were incubated with a solution of hDLL4-Fc (5 ug/ml) or mCD137-Fc (5 ug/ml) in PBS for 2 hours at 37 degrees C. Plates were then used to differentiate MSCs.
Functional Assay
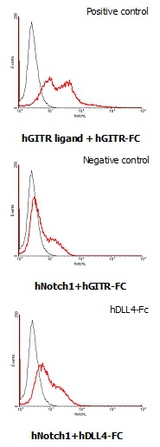
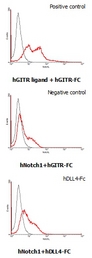
Interaction of human Notch1 with human DLL4. HEK293 cells transfected with a human Notch1 or a human GITR ligand expressing vector were incubated with 25 ug/ml of human GITR-Fc or human DLL4-Fc FITC conjugate for DLL4-Fc binding.
Functional Assay
Adipogenesis inhibition of 3T3L-1 cells.
Functional Assay
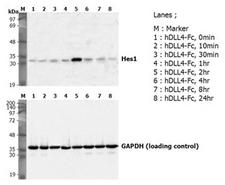
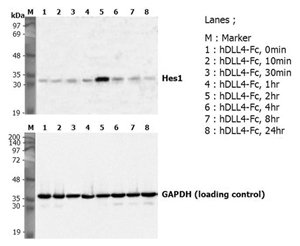
Induction of Hes-1 with the treatment of recombinant human DLL4-Fc (Prod. No. AG-40A-0077). A mouse preadpipocyte cell line, 3T3L1, was stimulated with 5 ug/ml of human DLL4-Fc as in indicated time points and each cell lysate was prepared and subjected to western blot by using anti-mouse Hes1 or GAPDH.
Functional Assay

Adipogenesis inhibition of 3T3L-1 cells. 3T3L-1 Cells(mouse pre-adipocyte cells) were maintained in DMEM, supplemented with 10% fetal bovine serum and penicillin-streptomycin. For differentiation of 3T3L-1 Cells, 3T3L-1 Cells were cultured in adipogenic medium which was growth medium supplemented with 1 uM Dexamethasone, 0.5mM IBMX, 10 ug/ml lnsulin (day 0). Medium was changed every 2 days. Staining with Oil Red O was typically performed on day 7. Cells were washed twice with PBS, fixed with 3.7% formalin, and stained with 0.5% filtered Oil Red O in propylene glycol. For negative controls, mouse TNF-alpha (20ng/ml) was added. Recombinant human DLL4-Fc (5 ug/ml) dissolved in DPBS was added to the differentiation medium. These plates were then used to differentiate 3T3L-1 Cells.
Functional Assay

Adipogenesis inhibition of MSCs. MSCs (Mesenchymal stem cells) were maintained in DMEM, supplemented with 10% fetal bovine serum, penicilin-streptomycin and glutamine. For differentiation of MSCs, MSCs were cultured in adipogenic medium which was growth medium supplemented with 1 uM Dexamethasone, 0.5mM IBMX, 10 ug/m lnsulin, 100 uM Indomethacin (day 1). Medium was changed every 3 days. Staining with Oil Red O was typically performed on day 30. For negative controls, TNF-alpha (20ng/ml) was added. To immobilize Notch ligands on the plastic surface of the culture plates, plates were incubated with a solution of hDLL4-Fc (5 ug/ml) or mCD137-Fc (5 ug/ml) in PBS for 2 hours at 37 degrees C. Plates were then used to differentiate MSCs.
Functional Assay

Interaction of human Notch1 with human DLL4. HEK293 cells transfected with a human Notch1 or a human GITR ligand expressing vector were incubated with 25 ug/ml of human GITR-Fc or human DLL4-Fc FITC conjugate for DLL4-Fc binding.
Functional Assay

Adipogenesis inhibition of 3T3L-1 cells.
Functional Assay

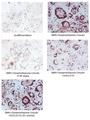
Induction of Hes-1 with the treatment of recombinant human DLL4-Fc (Prod. No. AG-40A-0077). A mouse preadpipocyte cell line, 3T3L1, was stimulated with 5 ug/ml of human DLL4-Fc as in indicated time points and each cell lysate was prepared and subjected to western blot by using anti-mouse Hes1 or GAPDH.
Functional Assay
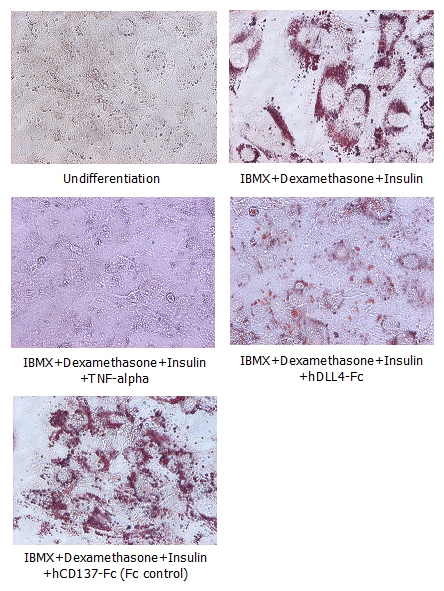
Adipogenesis inhibition of 3T3L-1 cells. 3T3L-1 Cells(mouse pre-adipocyte cells) were maintained in DMEM, supplemented with 10% fetal bovine serum and penicillin-streptomycin. For differentiation of 3T3L-1 Cells, 3T3L-1 Cells were cultured in adipogenic medium which was growth medium supplemented with 1 uM Dexamethasone, 0.5mM IBMX, 10 ug/ml lnsulin (day 0). Medium was changed every 2 days. Staining with Oil Red O was typically performed on day 7. Cells were washed twice with PBS, fixed with 3.7% formalin, and stained with 0.5% filtered Oil Red O in propylene glycol. For negative controls, mouse TNF-alpha (20ng/ml) was added. Recombinant human DLL4-Fc (5 ug/ml) dissolved in DPBS was added to the differentiation medium. These plates were then used to differentiate 3T3L-1 Cells.
Functional Assay
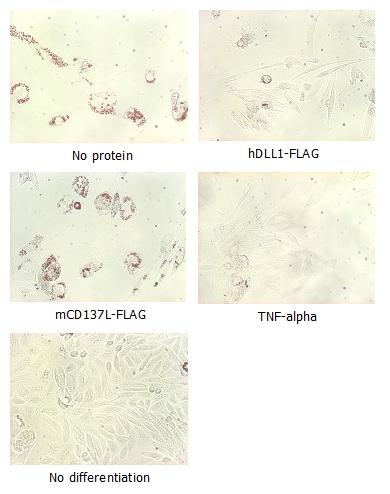
Adipogenesis inhibition of MSCs. MSCs (Mesenchymal stem cells) were maintained in DMEM, supplemented with 10% fetal bovine serum, penicilin-streptomycin and glutamine. For differentiation of MSCs, MSCs were cultured in adipogenic medium which was growth medium supplemented with 1 uM Dexamethasone, 0.5mM IBMX, 10 ug/m lnsulin, 100 uM Indomethacin (day 1). Medium was changed every 3 days. Staining with Oil Red O was typically performed on day 30. For negative controls, TNF-alpha (20ng/ml) was added. To immobilize Notch ligands on the plastic surface of the culture plates, plates were incubated with a solution of hDLL4-Fc (5 ug/ml) or mCD137-Fc (5 ug/ml) in PBS for 2 hours at 37 degrees C. Plates were then used to differentiate MSCs.
Functional Assay
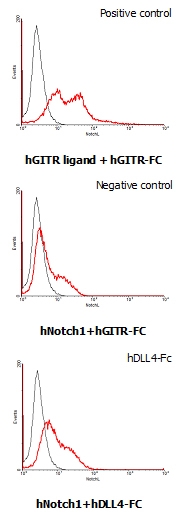
Interaction of human Notch1 with human DLL4. HEK293 cells transfected with a human Notch1 or a human GITR ligand expressing vector were incubated with 25 ug/ml of human GITR-Fc or human DLL4-Fc FITC conjugate for DLL4-Fc binding.
Functional Assay

Adipogenesis inhibition of 3T3L-1 cells.
Functional Assay

Induction of Hes-1 with the treatment of recombinant human DLL4-Fc (Prod. No. AG-40A-0077). A mouse preadpipocyte cell line, 3T3L1, was stimulated with 5 ug/ml of human DLL4-Fc as in indicated time points and each cell lysate was prepared and subjected to western blot by using anti-mouse Hes1 or GAPDH.
Functional Assay

Adipogenesis inhibition of 3T3L-1 cells. 3T3L-1 Cells(mouse pre-adipocyte cells) were maintained in DMEM, supplemented with 10% fetal bovine serum and penicillin-streptomycin. For differentiation of 3T3L-1 Cells, 3T3L-1 Cells were cultured in adipogenic medium which was growth medium supplemented with 1 uM Dexamethasone, 0.5mM IBMX, 10 ug/ml lnsulin (day 0). Medium was changed every 2 days. Staining with Oil Red O was typically performed on day 7. Cells were washed twice with PBS, fixed with 3.7% formalin, and stained with 0.5% filtered Oil Red O in propylene glycol. For negative controls, mouse TNF-alpha (20ng/ml) was added. Recombinant human DLL4-Fc (5 ug/ml) dissolved in DPBS was added to the differentiation medium. These plates were then used to differentiate 3T3L-1 Cells.
Functional Assay

Adipogenesis inhibition of MSCs. MSCs (Mesenchymal stem cells) were maintained in DMEM, supplemented with 10% fetal bovine serum, penicilin-streptomycin and glutamine. For differentiation of MSCs, MSCs were cultured in adipogenic medium which was growth medium supplemented with 1 uM Dexamethasone, 0.5mM IBMX, 10 ug/m lnsulin, 100 uM Indomethacin (day 1). Medium was changed every 3 days. Staining with Oil Red O was typically performed on day 30. For negative controls, TNF-alpha (20ng/ml) was added. To immobilize Notch ligands on the plastic surface of the culture plates, plates were incubated with a solution of hDLL4-Fc (5 ug/ml) or mCD137-Fc (5 ug/ml) in PBS for 2 hours at 37 degrees C. Plates were then used to differentiate MSCs.
Functional Assay

Interaction of human Notch1 with human DLL4. HEK293 cells transfected with a human Notch1 or a human GITR ligand expressing vector were incubated with 25 ug/ml of human GITR-Fc or human DLL4-Fc FITC conjugate for DLL4-Fc binding.
Functional Assay

Adipogenesis inhibition of 3T3L-1 cells.
Functional Assay

Induction of Hes-1 with the treatment of recombinant human DLL4-Fc (Prod. No. AG-40A-0077). A mouse preadpipocyte cell line, 3T3L1, was stimulated with 5 ug/ml of human DLL4-Fc as in indicated time points and each cell lysate was prepared and subjected to western blot by using anti-mouse Hes1 or GAPDH.
Popular DLL4 Proteins
Source:
Human
Tag:
Human IgG1 Fc
Source:
E. coli
Tag:
His, N-Terminal
Request SDS/MSDS
To request an SDS/MSDS form for this product, please contact our Technical Support department at:
Technical.Support@LSBio.com
Requested From: United States
Date Requested: 4/25/2024
Date Requested: 4/25/2024