Login
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Quick Order
Products
Antibodies
ELISA and Assay Kits
Research Areas
Infectious Disease
Resources
Purchasing
Reference Material
Contact Us
Locations
Orders Processing,
Shipping & Receiving,
Warehouse
2 Shaker Rd Suites
B001/B101
Shirley, MA 01464
Production Lab
Floor 6, Suite 620
20700 44th Avenue W
Lynnwood, WA 98036
Telephone Numbers
Tel: +1 (206) 374-1102
Fax: +1 (206) 577-4565
Contact Us
Additional Contact Details
Login
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Quick Order
| Catalog Number | Size | Price |
|---|---|---|
| LS-F19797-1 | 1 Plate | $783 |
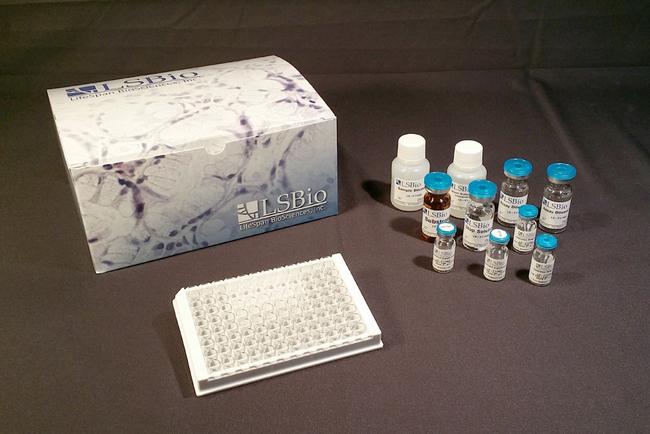
Rat C4.4A / LYPD3 (Custom ELISA) ELISA Kit - LS-F19797
Rat C4.4A / LYPD3 (Custom ELISA) ELISA Kit - LS-F19797
Description:
LS-F19797 is a 96-well enzyme-linked immunosorbent assay (ELISA) for the Quantitative detection of Rat C4.4A / LYPD3. It is based upon a Custom assay principle.
All LSBio Custom kits have been predesigned and all necessary components, including antibodies and standards have been identified.
Upon receiving a custom kit order, the kit will be assembled and quality control tested before being shipped out. Kit assembly and
testing typically takes 4 to 6 weeks. In most cases the final kit is based on the Sandwich assay principle, with a few being
EIA Competition based. Specifications such as Range, Sensitivity, and Precision are defined upon completion. In the event that the
custom kit cannot be successfully developed with 6 weeks of the order date, the customer will be notified and the order canceled at
no cost.
Toll Free North America
 206-374-1102
206-374-1102
For Research Use Only
Overview
Description:
LS-F19797 is a 96-well enzyme-linked immunosorbent assay (ELISA) for the Quantitative detection of Rat C4.4A / LYPD3. It is based upon a Custom assay principle.
All LSBio Custom kits have been predesigned and all necessary components, including antibodies and standards have been identified.
Upon receiving a custom kit order, the kit will be assembled and quality control tested before being shipped out. Kit assembly and
testing typically takes 4 to 6 weeks. In most cases the final kit is based on the Sandwich assay principle, with a few being
EIA Competition based. Specifications such as Range, Sensitivity, and Precision are defined upon completion. In the event that the
custom kit cannot be successfully developed with 6 weeks of the order date, the customer will be notified and the order canceled at
no cost.
Specifications
Type
Custom ELISA kit
Target
C4.4A / LYPD3
Synonyms
LYPD3 | C4.4A | MIG-C4 | LY6/PLAUR domain containing 3
Reactivity
Rat
Kit Components
Typical components include:
- Coated 96-well Strip Plate
- Standard (Lyophilized)
- Sample Diluent
- Assay Diluent A
- Assay Diluent B
- Detection Reagent A
- Detection Reagent B
- TMB Substrate
- Stop Solution
- Adhesive Plate Sealers
- Wash Buffer (25x)
- (See Datasheet for specific volumes supplied)
Manual
Format
96-Well Strip Plate
Detection
Colorimetric - 450nm (TMB)
Measurement
Quantitative
Detection Range
To be determined
Sensitivity
To be determined
Storage
Short term: 4°C; Long term: see manual.
Restrictions
For research use only. Intended for use by laboratory professionals.
Guarantee
This ELISA Kit carries the LSBio 100% Guarantee
About C4.4A / LYPD3
Publications (0)
Customer Reviews (0)
Images




Request SDS/MSDS
To request an SDS/MSDS form for this product, please contact our Technical Support department at:
Technical.Support@LSBio.com
Requested From: United States
Date Requested: 4/25/2024
Date Requested: 4/25/2024









