order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Locations
Orders Processing,
Shipping & Receiving,
Warehouse
2 Shaker Rd Suites
B001/B101
Shirley, MA 01464
Production Lab
Floor 6, Suite 620
20700 44th Avenue W
Lynnwood, WA 98036
Telephone Numbers
Tel: +1 (206) 374-1102
Fax: +1 (206) 577-4565
Contact Us
Additional Contact Details
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
order histories, retained contact details for faster checkout, review submissions, and special promotions.
BID
BH3 interacting domain death agonist
Apoptosis plays a major role in normal organism development, tissue homeostasis, and removal of damaged cells. Disruption of this process has been implicated in a variety of diseases such as cancer. The Bcl-2 family of proteins is comprised of critical regulators of apoptosis that can be divided into two classes: those that inhibit apoptosis and those that promote cell death. Bid, a pro-apoptotic Bcl-2 family member, is cleaved by caspase-8 in response to apoptotic signals, exposing the Bcl-2 homology 3 (BH3) domain which is normally buried in the full-length protein. The cleaved complex is myristoylated and translocated to the mitochondrial membrane where it may induce mitochondrial Bax and Bak to oligomerize.
| Gene Name: | BH3 interacting domain death agonist |
| Family/Subfamily: | Apoptosis |
| Synonyms: | BID, Apoptic death agonist, BID isoform L(2), BID isoform Si6, Bh3, FP497, p22 BID, BID isoform ES(1b), Desmocollin type 4, Human BID coding sequence |
| Target Sequences: | NM_001196 NP_001187.1 P55957 |

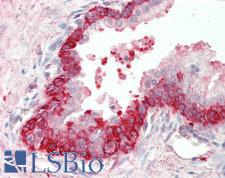

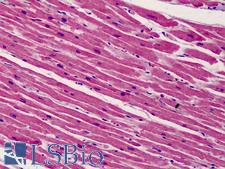
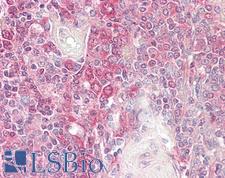
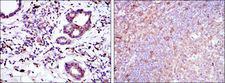
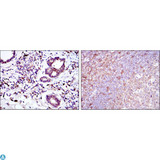
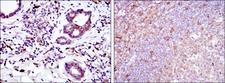

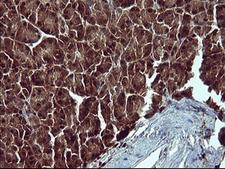
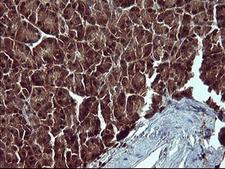
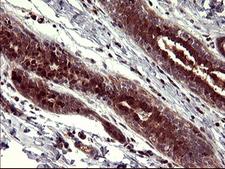
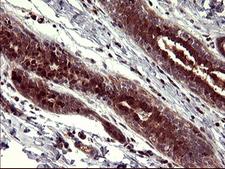
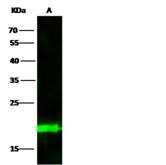
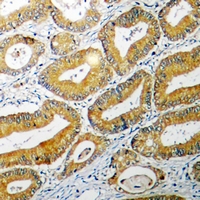
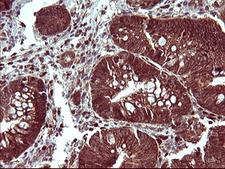
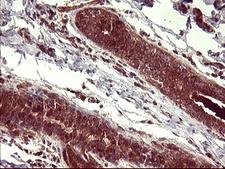
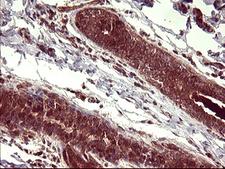
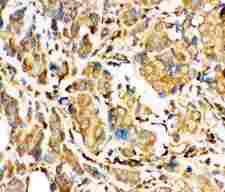
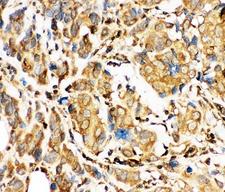
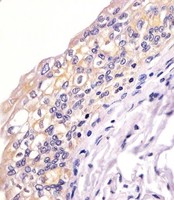
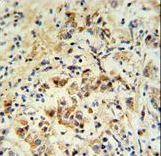
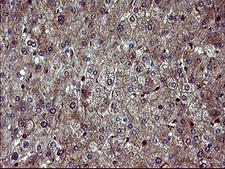
If you do not find the reagent or information you require, please contact Customer.Support@LSBio.com to inquire about additional products in development.










