Login
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Quick Order
Products
Antibodies
ELISA and Assay Kits
Research Areas
Infectious Disease
Resources
Purchasing
Reference Material
Contact Us
Locations
Orders Processing,
Shipping & Receiving,
Warehouse
2 Shaker Rd Suites
B001/B101
Shirley, MA 01464
Production Lab
Floor 6, Suite 620
20700 44th Avenue W
Lynnwood, WA 98036
Telephone Numbers
Tel: +1 (206) 374-1102
Fax: +1 (206) 577-4565
Contact Us
Additional Contact Details
Login
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Forgot password?
Registration enables users to use special features of this website, such as past
order histories, retained contact details for faster checkout, review submissions, and special promotions.
order histories, retained contact details for faster checkout, review submissions, and special promotions.
Quick Order
| Catalog Number | Size | Price |
|---|---|---|
| LS-B2303-0.1 | 0.1 ml | $460 |


1 of 2
2 of 2
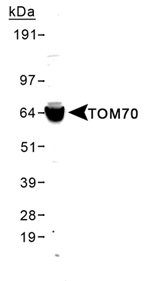
IHC‑plus™ Polyclonal Rabbit anti‑Human TOMM70A Antibody (aa1‑100, IHC, WB) LS‑B2303
IHC‑plus™ Polyclonal Rabbit anti‑Human TOMM70A Antibody (aa1‑100, IHC, WB) LS‑B2303
Note: This antibody replaces LS-C33974, LS-C90607
Antibody:
TOMM70A Rabbit anti-Human Polyclonal (aa1-100) Antibody
Application:
IHC, IHC-P, WB
Reactivity:
Human, Mouse, Rat
Format:
Unconjugated, Unmodified
Toll Free North America
 206-374-1102
206-374-1102
For Research Use Only
Overview
Antibody:
TOMM70A Rabbit anti-Human Polyclonal (aa1-100) Antibody
Application:
IHC, IHC-P, WB
Reactivity:
Human, Mouse, Rat
Format:
Unconjugated, Unmodified
Specifications
Description
TOMM70A antibody LS-B2303 is an unconjugated rabbit polyclonal antibody to TOMM70A (aa1-100) from human. It is reactive with human, mouse and rat. Validated for IHC and WB. Tested on 20 paraffin-embedded human tissues.
Host
Rabbit
Reactivity
Human, Mouse, Rat
(tested or 100% immunogen sequence identity)
Clonality
Polyclonal
Conjugations
Unconjugated
Purification
Protein A purified
Modifications
Unmodified
Immunogen
A synthetic peptide made to a region within residues 1-100 of the human protein. [Swiss-Prot# O94826]
Epitope
aa1-100
Specificity
TOM70.
Applications
- IHC
- IHC - Paraffin (2.5 - 5 µg/ml)
- Western blot
|
Performing IHC? See our complete line of Immunohistochemistry Reagents including antigen retrieval solutions, blocking agents
ABC Detection Kits and polymers, biotinylated secondary antibodies, substrates and more.
|
Usage
Immunohistochemistry: LS-B2303 was validated for use in immunohistochemistry on a panel of 21 formalin-fixed, paraffin-embedded (FFPE) human tissues after heat induced antigen retrieval in pH 6.0 citrate buffer. After incubation with the primary antibody, slides were incubated with biotinylated secondary antibody, followed by alkaline phosphatase-streptavidin and chromogen. The stained slides were evaluated by a pathologist to confirm staining specificity. The optimal working concentration for LS-B2303 was determined to be 2.5-5 ug/ml.
Presentation
PBS
Storage
Aliquot and store at -20°C or -80°C. Avoid freeze-thaw cycles.
Restrictions
For research use only. Intended for use by laboratory professionals.
About TOMM70A
Validation
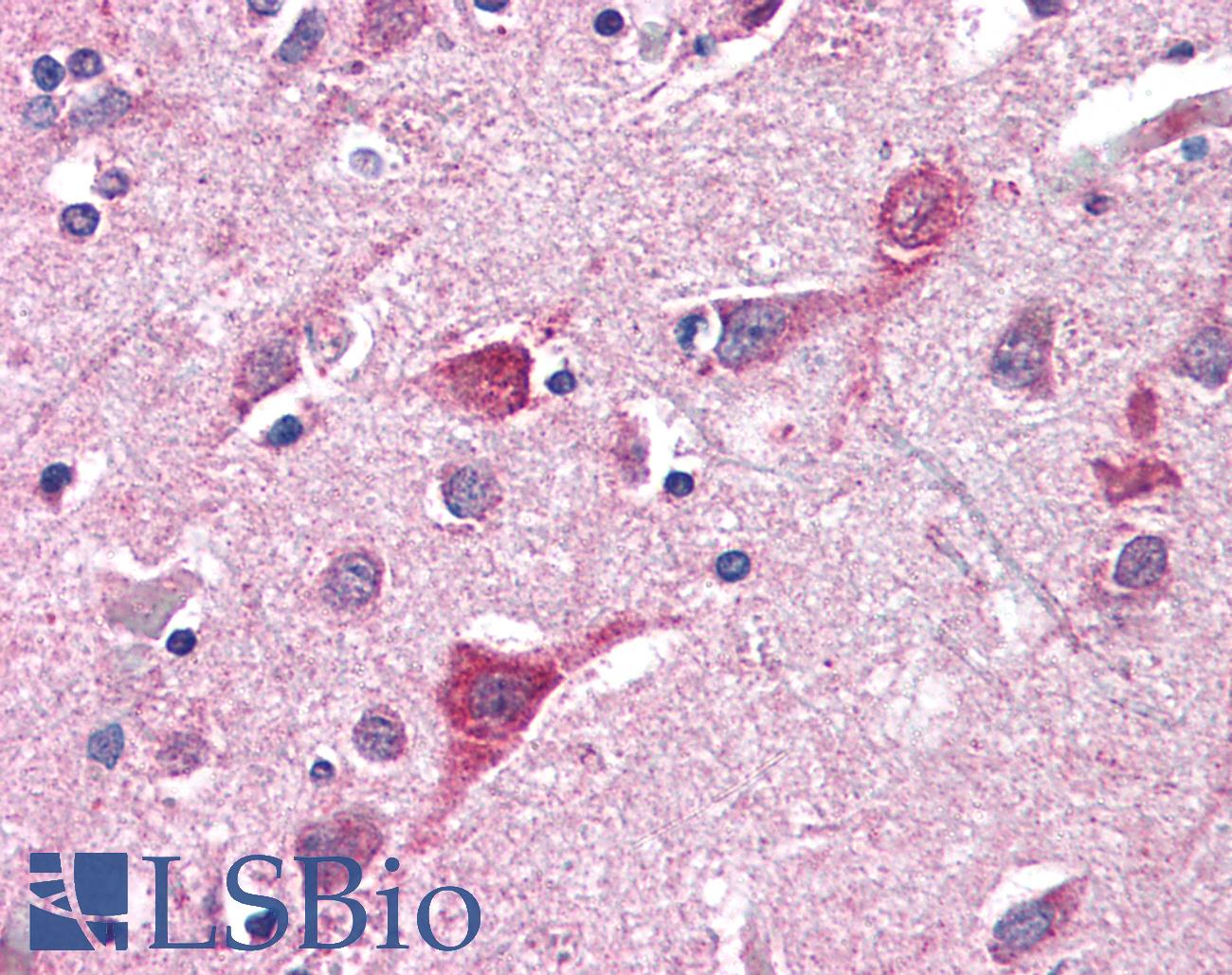
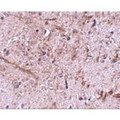
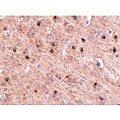
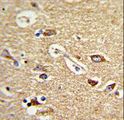
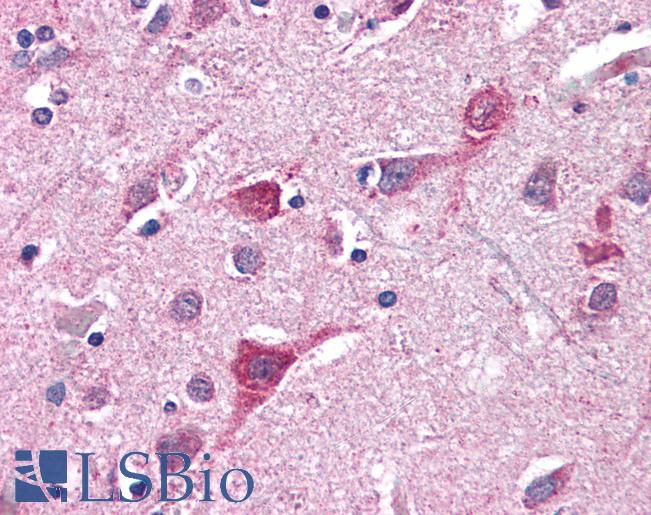
Anti-TOMM70A antibody IHC of human brain, cortex. Immunohistochemistry of formalin-fixed, paraffin-embedded tissue after heat-induced antigen retrieval. Antibody concentration 5 ug/ml. This image was taken for the unconjugated form of this product. Other forms have not been tested.
Anti-TOMM70A antibody IHC of human brain, cortex. Immunohistochemistry of formalin-fixed, paraffin-embedded tissue after heat-induced antigen retrieval. Antibody concentration 5 ug/ml. This image was taken for the unconjugated form of this product. Other forms have not been tested.
See More About...
LSBio Ratings
IHC-plus™ TOMM70A Antibody (aa1-100) for IHC, WB/Western LS-B2303 has an LSBio Rating of
Laboratory Validation Score (4)
Learn more about The LSBio Ratings Algorithm
Publications (0)
Customer Reviews (0)
Featured Products
Species:
Human
Applications:
IHC, Immunofluorescence, Western blot, Immunoprecipitation, Flow Cytometry, ELISA
Species:
Human, Mouse, Rat
Applications:
IHC, Immunofluorescence, Western blot, ELISA
Species:
Human, Mouse, Rat
Applications:
IHC, Immunofluorescence, Western blot, ELISA
Species:
Human
Applications:
IHC, IHC - Paraffin, Western blot, Flow Cytometry
Request SDS/MSDS
To request an SDS/MSDS form for this product, please contact our Technical Support department at:
Technical.Support@LSBio.com
Requested From: United States
Date Requested: 4/24/2024
Date Requested: 4/24/2024